A method for estimating electron affinities is to extrapolate Z eff values for atoms and ions that
Question:
A method for estimating electron affinities is to extrapolate Zeff values for atoms and ions that contain the same number of electrons as the negative ion of interest. Use the data in the table to answer the questions that follow.
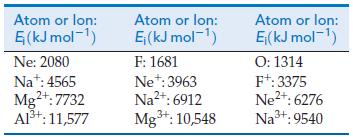
(a) Estimate the electron affinity of F, and compare it with the experimental value.
(b) Estimate the electron affinities of O and N.
(c) Examine your results in terms of penetration and screening.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:





