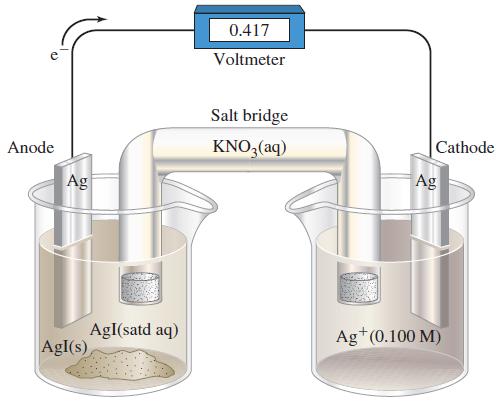
Assume that the volume of each solution in Figure 19-22 is 100.0 mL. The cell is operated
Question:
Assume that the volume of each solution in Figure 19-22 is 100.0 mL. The cell is operated as an electrolytic cell, using a current of 0.500 A. Electrolysis is stopped after 10.00 h, and the cell is allowed to function as a voltaic cell. What is Ecell at this point?
Figure 19-22
Transcribed Image Text:
Anode Ag AgI(s) AgI(satd aq) 0.417 Voltmeter Salt bridge KNO3(aq) Cathode Ag Ag+ (0.100 M)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To answer this question we need to first calculate the amount of silver deposited on the cathode during electrolysis We can use Faradays first law of ...View the full answer

Answered By

ANUJ SHARMA
I am an Expert-Q&A in Finance and Accountancy, providing solutions for Ph.D. and Masters level finance problems.
Throughout my tutoring career, my primary focus is on the intellectual enrichment of the students.
By providing them simplified explanations, and high-quality academic material for complex concepts related to theoretical and applied fields.
I believe in assisting the students in their explorations and endeavors that push them to the new extremes.
I Solve Applied and Theoretical (Academic and Real World) related to Accounting (Management, Corporate (U.S. GAAP, SEC)) and Finance (Financial Markets both Money Market and Capital Market, Capital Rationing, Valuation, Working Capital Management, Trading FX, Commodities, Equity, Options, Index, Etc.)
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The Stratton Township Park is located on a piece of property that contains two golf courses, a swimming pool, and 800 acres of woods and open spaces. Three years ago, the Stratton Park Department...
-
A box is separated by a partition into two parts of equal volume. The left side of the box contains 500 molecules of nitrogen gas; the right side contains 100 molecules of oxygen gas. The two gases...
-
EXECUTIVE SUMMARY In this report, there is an in-depth analysis of logistics revolving around a case study based on an article by Emmanuel Hassoun, a supply chain professional and Pierre Mawet, a...
-
What are the main advantages and disadvantages of using 360 degree appraisal?
-
Zanda Drug Corporation buys three chemicals that are processed to produce two types of analgesics used as ingredients for popular over-the-counter drugs. The purchased chemicals are blended for two...
-
Figure 4-38 includes an EER diagram for a medium-size software vendor. Transform the diagram into a relational schema that shows referential integrity constraints (see Figure 4-5 for an example of...
-
Do you offer free information? Avoid organizations that charge for information or demand details about your problem first.
-
A study of cigarette demand resulted in the following logarithmic regression equation: Here, Q denotes annual cigarette consumption, P is the average price of cigarettes, Y is per capita income, A is...
-
Cash Receipts The sales budget for Perrier Inc. is forecasted as follows: Month Sales Revenue Mary $120.000 Chine 160.000 My 130.000 August 120,000 To prepare a cash budget, the company must...
-
You prepare 1.00 L of a buffer solution that is 1.00 M NaH 2 PO 4 and 1.00 M Na 2 HPO 4 . The solution is divided in half between the two compartments of an electrolytic cell. Both electrodes used...
-
It is sometimes possible to separate two metal ions through electrolysis. One ion is reduced to the free metal at the cathode, and the other remains in solution. In which of these cases would you...
-
Use Green's Theorem to evaluate the line integral. Orient the curve counterclockwise unless otherwse indicated. \(\oint_{C} 5 y d x+2 x d y\), where \(C\) is the triangle with vertices...
-
A storeroom is used to organize items stored in it on N shelves. Shelves are numbered from 0 to N-1. The K-th shelf is dedicated to items of only one type, denoted by a positive integer A[K]....
-
CASES CASE 10.1 Money in Motion Jake Nguyen runs a nervous hand through his once finely combed hair. He loosens his once perfectly knotted silk tie. And he rubs his sweaty hands across his once...
-
(3.8) Axiom, Definition of false false = true (3.9) Axiom, Distributivity of over : (pq) p=q
-
The board of directors of Unilever has been impressed by the presentation you did, and they further instructed you to conduct a more insightful investigation about the Sri Lankan market. They have...
-
The sample space listing the eight simple events that are possible when a couple has three children is {bbb, bbg, bgb, ogg, gbb, gbg, ggb, ggg}. After identifying the sample space for a couple having...
-
Senator Borman interjects the following comment after the statements by Senators Creighton and Long reported in Problems 2-8 and 2-9: "In fact, both of my esteemed colleagues are wrong, because an...
-
Give an example of transitory income. What effect does this income have on the marginal propensity to consume?
-
On July 1, 2010, Remington Chemical Company issued $4,000,000 face value, 10%,10-year bonds at $4,543,627.This price resulted in an 8% effective-interest rate on the bonds. Remington uses the...
-
Suppan Company sold $6,000,000, 9%, 20-year bonds on January 1, 2010. The bonds were dated January 1, 2010, and pay interest on January 1 and July 1. Suppan Company uses the straight-line method to...
-
Jinkens Corporation sold $4,000,000, 8%, 10-year bonds on January 1, 2010.The bonds were dated January 1, 2010, and pay interest on July 1 and January 1. Jinkens Corporation uses the straight-line...
-
Phantom Consulting Inc. is a small computer consulting business. The company is organized as a corporation and provides consulting services, computer system installations, and custom program...
-
Sam owns a 25% in Spade, LLC. In 2021, Spade reports $100,000 or ordinary income. What is Sams qualified business income (QBI) deduction? answer is 5,000 but please show how to get it
-
crane Inc. common chairs currently sell for $30 each. The firms management believes that it's share should really sell for $54 each. If the firm just paid an annual dividend of two dollars per share...

Study smarter with the SolutionInn App


