Estimate K at 100 C for the reaction 2 SO 2 (g) + O 2 (g)
Question:
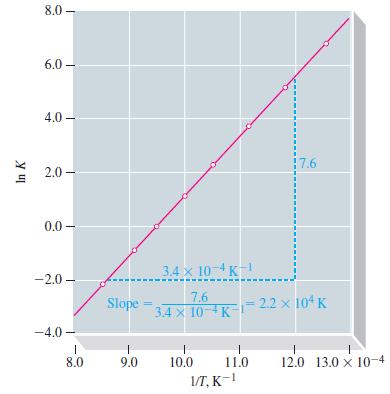
Estimate K at 100 °C for the reaction 2 SO2(g) + O2(g) Δ 2 SO3(g). Use data from Table 13.8 and Figure 13-10.
Table 13.8
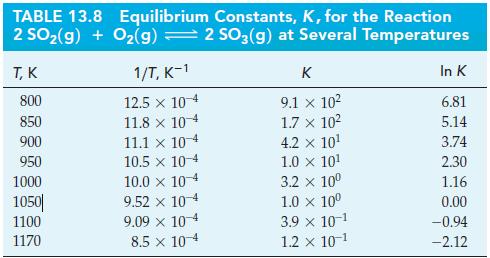
Figure 13-10
Transcribed Image Text:
TABLE 13.8 Equilibrium Constants, K, for the Reaction 2 SO₂(g) + O₂(g) 2 SO3(g) at Several Temperatures T, K 800 850 900 950 1000 1050 1100 1170 1/T, K-1 12.5 x 10-4 11.8 x 10-4 11.1 x 10-4 10.5 x 10-4 10.0 × 10-4 9.52 x 10-4 9.09 × 10-4 8.5 x 104 K 9.1 x 10² 1.7 x 10² 4.2 x 10¹ 1.0 x 10¹ 3.2 x 10⁰ 1.0 x 10⁰ 3.9 x 10-1 1.2 x 10-¹ In K 6.81 5.14 3.74 2.30 1.16 0.00 -0.94 -2.12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To estimate K at 100 C for the reaction 2 SO2g O2g 2 SO3g we can use the vant Hoff e...View the full answer

Answered By

Benard Ndini Mwendwa
I am a graduate from Kenya. I managed to score one of the highest levels in my BS. I have experience in academic writing since I have been working as a freelancer in most of my time. I am willing to help other students attain better grades in their academic portfolio. Thank you.
4.90+
107+ Reviews
240+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Estimate the temperature at which K = 5.8 x 10 -2 for the reaction in Example 13-12. Use data from Table 13.8 and Figure 13-10. (B) What is the value of K p for the reaction 2 SO 2 (g) + O 2 (g) ...
-
Use data from Table to estimate ÎH for the combustion of methane (CH4), as shown below: Table s | 14 39 95 45 72 1 1419 6847064968 77386 42222 34 985 0302 121 Si H C O 437 490 9 31222241122...
-
Carol Harris, Ph.D, CPA, is a single taxpayer and she lives at 674 Yankee Street, Durham, NC 27409. Her Social Security number is 793-52-4335. Carol is an Associate Professor of Accounting at a local...
-
Using result (8.9), find f(x) when f (x) is (a) x (b) 1/x 5 (c) 1 3x
-
In May 2014, the budget committee of Grand Stores assembles the following data in preparation of budgeted merchandise purchases for the month of June. 1. Expected sales: June $500,000, July $600,000....
-
In this exercise, we use multinomial logit to describe factors leading an individual to fall into one of three categories. Use data file \(r w m 88\) for this exercise. a. Create a variable called...
-
Define the rules that will cause the switch between main contexts and subcontexts in the aforementioned context hierarchy.
-
Michael Hauck claimed that he was discharged by his employer, Sabine Pilot Service, because he refused its direction to perform the illegal act of pumping the bilges of the employers vessel into the...
-
Problem 7-18 Activity-Based Costing and Bidding on Jobs [LO7-2, LO7-3, LO7-4] Mercer Asbestos Removal Company removes potentially toxic asbestos insulation and related products from buildings. There...
-
What must be the temperature if the following reaction has r G = -45.5 kJ mol -1 , r H = -24.8 kJ mol -1 , and r S = 15.2 J mol -1 K -1 ? Fe2O3(s) + 3 CO(g) 2 Fe(s) + 3 CO(g)
-
Use data from Appendix D to determine (a) r H, r S, and r G at 298 K and (b) K at 875 K for the water gas shift reaction, used commercially to produce H 2 (g): CO(g) + H 2 O(g) CO 2 (g) + H 2...
-
Finding implicit interest rates constructing amortization schedules. Herman Company purchased a plot of land for possible future development. The land had fair value of $86,000. Berman Company gave a...
-
How do marketers use new products to maintain and grow their market share? Your response must include a specific example of a company that successfully grew its business or attracted a new target...
-
How do you encourage cross-functional synergy within your organization to break down silos and facilitate innovative solutions to complex challenges ?
-
1. what is intended internal resource strategies. How do you plan to develop or acquire resources (tangible and/or intangible) that would generate core competencies? What are examples of resource...
-
1. What is a forward contract? 2. Why do you need fx swap ? 3. If the rate is usd to cad, then you multiple by the rate. If converting USD to CAD. If the rate is CAD to USD, then you have to divide...
-
1. Watch the video on Taylorism. Why do you think Frederick Taylor's ideas were important at that particular point in history, the early 1900s? Explain. 2. How were the Hawthorne studies a major...
-
A 4-kVA, 2,300/230-V rms transformer has an equivalent impedance of 210 on the primary side. If the transformer is connected to a load with 0.6 power factor leading, calculate the input impedance.
-
You are standing at x = 9.0 km and your assistant is standing at x = 3.0 km. Lightning bolt 1 strikes at x = 0 km and lightning bolt 2 strikes at x = 12.0 km. You see the flash from bolt 2 at t = 10...
-
Denominator-level choices, changes in inventory levels, effect on operating income. Koshu Corporation is a manufacturer of computer accessories. It uses absorption costing based on standard costs and...
-
Effects of denominator-level choice. The Shen Company is a manufacturer of MP3 players. It installed standard costs and a flexible budget on January 1, 2009. The president has been pondering how...
-
Downward demand spiral. Network Company is a large manufacturer of optical storage systems based in Arizona. Its practical annual capacity is 7,500 units, and, for the past few years, its budgeted...
-
Rotan, Inc. purchased a van on January 1, 2018, for $800,000. Estimated life of the van was five years, and its estimated residual value was $96,000. Rotan uses the straightline method of...
-
If at the beginning of a period, you buy a share of stock for $49, then receive a dividend of $3, and finally sell the stock for $51, what was your holding period return? 9.3% 10.2% 14.8% 16.3%
-
The cumulative effect of most changes in accounting principle is reported: Multiple Choice In the balance sheet accounts affected. In the income statement before income from continuing operations. In...

Study smarter with the SolutionInn App


