From the data given in Exercise 72, estimate a value of r S at 298 K
Question:
From the data given in Exercise 72, estimate a value of ΔrS° at 298 K for the reaction
![]()
Exercise 72
Sodium carbonate, an important chemical used in the production of glass, is made from sodium hydrogen carbonate by the reaction
![]()
Data for the temperature variation of K for this reaction are K = 1.66 x 10-5 at 30 °C; 3.90 x 10-4 at 50 °C; 6.27 x 10-3 at 70 °C; and 2.31 x 10-1 at 100 °C.
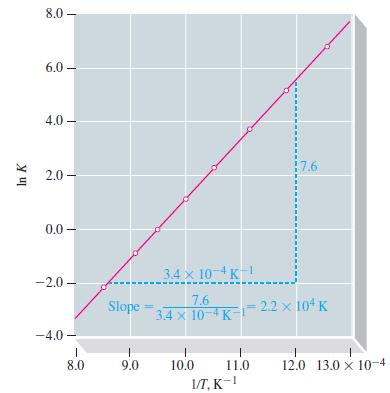
(a) Plot a graph similar to Figure 13-10, and determine ΔrH° for the reaction.
(b) Calculate the temperature at which the total gas pressure above a mixture of NaHCO3(s) and Na2CO3(s) is 2.00 bar.
Figure 13-10
Step by Step Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:




