We saw that electronegativity differences determine whether bond dipoles exist in a molecule and that molecular shape
Question:
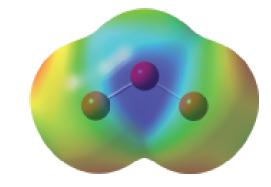
We saw that electronegativity differences determine whether bond dipoles exist in a molecule and that molecular shape determines whether bond dipoles cancel (nonpolar molecules) or combine to produce a resultant dipole moment (polar molecules). Thus, the ozone molecule, O3, has no bond dipoles because all the atoms are alike. Yet, O3 does have a resultant dipole moment: μ = 0.534 D. The electrostatic potential map for ozone is shown below. Use the electrostatic potential map to decide the direction of the dipole. Using the ideas of delocalized bonding in molecules, can you rationalize this electrostatic potential map?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:





