Formic acid, HCHO 2 , is used to make methyl formate (a fumigant for dried fruit) and
Question:
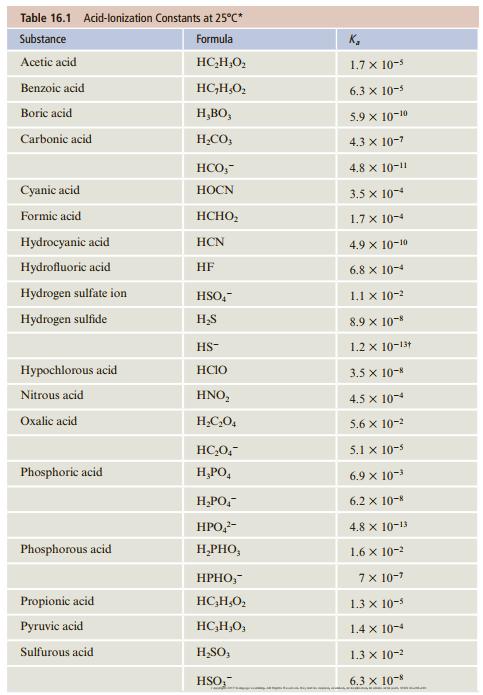
Formic acid, HCHO2, is used to make methyl formate (a fumigant for dried fruit) and ethyl formate (an artificial rum flavor). What is the pH of a 0.12 M solution of formic acid? What is the degree of ionization of HCHO2 in this solution? See Table 16.1 for Ka.

Transcribed Image Text:
Table 16.1 Acid-lonization Constants at 25°C* Substance Formula K, Acetic acid HC,H,O, 1.7 x 10-5 Benzoic acid HC,H,O, 6.3 x 10-5 Boric acid H,BO, 5.9 x 10-10 Carbonic acid H,CO, 4.3 x 10-7 HCO,- 4.8 x 10-11 Cyanic acid HOCN 3.5 x 10-4 Formic acid HCHO, 1.7 x 10-4 Hydrocyanic acid HCN 4.9 x 10-10 Hydrofluoric acid HF 6.8 x 10-4 Hydrogen sulfate ion HSO,- 1.1 x 10-2 Hydrogen sulfide H,S 8.9 x 10- 1.2 x 10-13 HS- Hypochlorous acid HCIO 3.5 x 10-8 Nitrous acid HNO, 4.5 x 10-4 Oxalic acid H,C,O, 5.6 x 10-2 HC,0,- 5.1 x 10-5 Phosphoric acid H,PO, 6.9 x 10-3 H,PO,- 6.2 x 10-8 HPO,- 4.8 x 10-1 Phosphorous acid H,PHO, 1.6 x 10-2 НРНО,- 7x 10-7 Propionic acid HC,H,O, 1.3 x 10-5 Рyruvic acid HC,H,O, 1.4 x 10-4 Sulfurous acid H,SO, 1.3 x 10-2 HSO, 6.3 x 10-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
To solve assemble a table of starting change and equilibrium concentrations ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Formic acid, HCHO2, is used to make methyl formate (a fumigant for dried fruit) and ethyl formate (an artificial rum flavor). What is the pH of a 0.10 M solution of formic acid? What is the degree of...
-
A 1.24-g sample of benzoic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.180 M NaOH. What was the pH of the solution when the equivalence point was...
-
A 0.400-g sample of propionic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.150 M NaOH. What was the pH of the solution when the equivalence point was...
-
If r = (t, t 2 , t 3 ), find r(t) and r(t).
-
The importance of having employees with a healthy work-life balance has been recognized by U.S. companies for decades. Many business schools offer courses that assist MBA students with developing...
-
Figure shows the first four peaks of the x-ray diffraction pattern for copper, which has an FCC crystal structure; monochromatic x-radiation having a wavelength of 0.1542 nm was used. (a) Index...
-
Prove that the inverse hyperbolic functions are the following logarithms: a. \(\cosh ^{-1} x=\ln \left(x+\sqrt{x^{2}-1} ight)\). b. \(\tanh ^{-1} x=\frac{1}{2} \ln \frac{1+x}{1-x}\).
-
Sara Casper, a recent graduate of Rollings accounting program, evaluated the operating performance of Klumpe Companys six divisions. Sara made the following presentation to the Klumpe board of...
-
Recently, several banks have started offering customers remote deposit capture. With this new service, customers do not have to physically go to banks or ATM machines to deposit checks. Instead, they...
-
Compute the missing amounts In the separate Income statements A, B, and C. Sales 24 21,200 92,000 Cost of goods sold 40,800 30,800 Gross profit 36,200 12,300 Expenses 6,400 Net income 13,800 21,800...
-
Boric acid, B(OH) 3 , is used as a mild antiseptic. What is the pH of a 0.015 M aqueous solution of boric acid? What is the degree of ionization of boric acid in this solution? The hydronium ion...
-
C 6 H 4 NH 2 COOH, para-aminobenzoic acid (PABA), is used in some sunscreen agents. Calculate the concentrations of hydronium ion and para-aminobenzoate ion, C 6 H 4 NH 2 COO - , in a 0.080 M...
-
____ ____ is a measure of how effectively a company engages with its various online stakeholders in a mutually beneficial exchange of information.
-
What types of information are particularly important to an international firm?
-
How are operations management, productivity, and quality interrelated?
-
What are the basic similarities and differences between production management and service operations management?
-
Why are services most closely associated with developed, industrialized economies?
-
What is fair trade? Who benefits from it?
-
If f(x) = x3 - 4/x, find the following. (a) f(-1/2) (b) f(2) (c) f(-2)
-
Explain briefly what is meant by electronic data interchange (EDI). How does EDI affect a companys audit trail?
-
For the decomposition of one mole of nitrosyl chloride, H = 38 kJ. NOCl(g) NO(g) + 1 / 2 Cl2(g) The activation energy for this reaction is 100 kJ. a. Is this reaction exothermic or endothermic? b....
-
Given the following mechanism for a chemical reaction: H2O2+ I H2O + IO H2O2+ IO H2O + O2 + I a. Write the overall reaction. b. Identify the catalyst and the reaction intermediate. c. With the...
-
Rate constants for reactions often follow the Arrhenius equation. Write this equation and then identify each term in it with the corresponding factor or factors from collision theory. Give a physical...
-
The pension plan assets balance is reported Multiple choice question. in the disclosure notes. in the income statement. in the balance sheet. in the statement of cash flows
-
Entering notes is the main function of notation apps. Higher price solutions like Finale and Sibelius offer a lot of entry options. Which item(s) below are not included? Group of answer choices...
-
Companies with relatively high assets-to-sales ratios (= A*/S) require a relatively large amount of new assets for any given increase in sales; but that doesn't always imply that they have a greater...

Study smarter with the SolutionInn App


