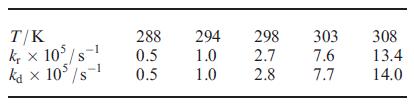
The rate constants for racemization (kr) and dissociation (k d ) of [FeL 3 ] 4 (H
Question:
The rate constants for racemization (kr) and dissociation (kd) of [FeL3]4− (H2L = 26.12) at several temperatures, T, are given in the table.
(a) Determine ![]() for each reaction.
for each reaction.
(b) What can you deduce about the mechanism of racemization?
Transcribed Image Text:
ΔΗ AH* and AS*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To determine the activation energy H and the entropy of activation S for the racemization reaction of FeL34 we can use the Arrhenius equation k A expH ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The gas mileage of a subcompact car is given by m = -0.04x 2 + 3.6x - 49, where x = the speed in miles per hour and m is given in miles per gallon. a) What is the gas mileage for a speed of 25 miles...
-
In order to solve this problem, you must read the description of the Hammett Ï, Ï treatment given in Chapter 17, Problem 76. When the rate constants for the hydrolysis of several morpholine...
-
Most humans are infected with herpes simplex virus-1 (HSV-1), the causative agent of cold sores. The HSV-1 genome comprises about I 00 genes, most of which are expressed in infected host cells at the...
-
For the curve defined by F(t) = (etcos(t), e* sin(t)) find the unit tangent vector, unit normal vector, normal acceleration, and tangential acceleration at 5A t 6 T N (5r) = 6 5T 6 aN ||
-
Keystone, LLC, owns a peach processing plant. Last month, the plant generated the following information: peaches processed, 60,000 pounds; direct materials, $6,200; direct labor, $12,500; variable...
-
Find the transfer function, G(s) = X(s)/E a (s), for the system shown in Figure P2.33. + e(n Motor N = 10 J= 1 kg-m? 3 D=1 N-m-s/rad N2 = 20 Radius = 2 m Ideal gear 1:1 M=I'kg For the motor: J. =1...
-
What is an impairment of a tangible capital asset? How is a depreciable capital asset impairment recorded in the financial statements?(Appendix)
-
Southern Exposure Ltd. begins operations on January 2, 2016. During the year, the following transactions affect shareholders equity: 1. Southern Exposures articles of incorporation authorize the...
-
International Finance Problem Set on Parity Conditions 1.The price of a tall vanilla latte at Starbucks in New York is $3.25 while in Rome it is 3. The current spot exchange rate is $1.1050/.Assuming...
-
Two twist mechanisms for the rearrangement of are shown in Fig. 26.9. The initial diagrams in (a) and (b) are identical; confirm that the enantiomers formed in (a) and (b) are also identical. Fig....
-
The structure of cisplatin is shown below: Despite its success as an anticancer drug, the mechanism by which the drug targets DNA in the body is not fully understood, although it is known that the...
-
Explain what we mean by "conditional probability."
-
Dionex Corporation, a leader in the development and manufacturing of ion chromography systems, reported EPS of $2.02 in 1993 and paid no dividends. These earnings are expected to grow at 14% a year...
-
Company produces 99,000 units and sells 100,000 The full unit costs are: DM$ 8 DL4 VOH17 FOH15 Which method of inventory costing will be higher and by how much?
-
The weight of a concrete column is 10.3 kN with a density of 2.30 g/cm3 . (a) Convert the weight from kN to N and the density from g/cm3 to kg/m3 . Note: 100 cm = 1 m. (b) Determine the mass of the...
-
Greenwood Ltd., one of the largest commercial builders in the city, has decided to purchase a new piece of equipment due to a very strong order book.You have just been hired as their new financial...
-
A bank customer wants to make a payment to credit card from his/her savings account. The amount payable is $1000. Describe steps of the transaction
-
Let An, n = 1, 2,..., be independent events in the probability space ((, A, P), and suppose that lim n( An exits, call it A. Then show that P(A) = 0 or P(A) = 1.
-
CLASS PERIO Solving Linear Equations: Variable on Both Sides Solve each equation. 1) 6r+ 7 = 13 + 7r 3) -7x-3x+2=-8x-8 5)-14 +66+7-26=1+5b 7) n-3n = 14-4n 2) 13-4x=1-x 4)-8-x= x - 4x 6)n+2=-14-n 8)...
-
Predict the product of each of the following reactions: (a) (b) [1] -NH2 -H20 [H*] H2N-NH2 -H20
-
Identify the reactants that you would use to make each of the following compounds: (a) (b) NH2
-
Draw a plausible mechanism for each of the following reactions: (a) (b) [H2SO4] Et,NH 'N' -H20 [H2SO4] Me-NH -H20
-
Net Gain or Loss Lee Company has a defined benefit pension plan. During 2 0 1 8 , for the first time, Lee experienced a difference between its expected and actual projected benefit obligation. At the...
-
a . With the aid of examples, discuss the differences between diversifiable and non - diversifiable risks. ( 5 marks ) b . Distinguish between loss control and premiums and show their relationship. (...
-
XYZ Corporation's balance sheet on December 31, 2023, is provided below: Item Amount ($) Accounts Payable 1,600,000 Notes Payable 1,580,000 Accrued Expenses 1,460,000 Total Current Liabilities ?...

Study smarter with the SolutionInn App


