In a kinetic study of the reaction the following rate data were obtained. Write a rate law
Question:
In a kinetic study of the reaction
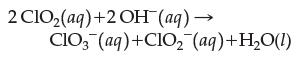
the following rate data were obtained. Write a rate law complete with proper values for the orders. What is the overall order of the reaction?![Experiment 1 23 2 [C10] 0.060 M 0.020 M 0.020 M [OH-] 0.030 M 0.030 M 0.090 M Rate (M/s) 0.024 84 0.002 76](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/5/9/10265704f1e898191701859100897.jpg)
Transcribed Image Text:
2 C10₂(aq)+ 2 OH (aq) → ClO3(aq)+CIO₂ (aq) +H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine the rate law for the given reaction and the overall order you can use the provided rate ...View the full answer

Answered By

Lucy Lagamayo
I taught Japanese professionals online as ESL teacher and as Assistant Language Teacher in a public school in Japan.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
In a kinetic study of the reaction 2A(g) + B(g) P(g) the following rate data were obtained. Write the rate law with proper orders. Give the overall order of the reaction. Finally, state what this...
-
In a kinetic study of the reaction 2NO(g) + O2(g) 2NO2(g) the following data were obtained for the initial rates of disappearance of NO: Obtain the rate law. What is the value of the rate constant?...
-
Consider the following reaction between mercury (II) chloride and oxalate ion: 2 HgCI2(aq) + C2O42- (aq) 2 CIË (aq) + 2 CO2(g) + Hg2CI2(s) The initial rate of this reaction was determined for...
-
True or False? Azure storage is used by both Infrastructure as a Service ( ( laaS ) ) virtual machines, and Platform as a Service ( ( PaaS ) ) cloud services. True False
-
Determining Financial Statement Effects of Adjusting Entries Refer to P4-3. Required: 1. Indicate whether each transaction relates to a deferred revenue, deferred expense, accrued revenue, or accrued...
-
Suppose that an increase in income transfers rather than government spending was the preferred policy for stimulating the economy depicted in Figure 11.5. By how much would transfers have to increase...
-
In what way might the IASBs approach to standard setting be relevant for the FASB? LO4
-
Reconsider the data presented in Exercise 6.21. (a) Rework both parts (a) and (b) of Exercise 6.21 using the x and s charts. (b) Does the s chart detect the shift in process variability more quickly...
-
The Fed decides to sell $20,000 of government bonds to Goldman Sachs. Using T-accounts show the complete transaction. Did the money supply increase or decrease? Explain.
-
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction? Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start...
-
A student measures the rate of a reaction by measuring how much the concentration of one of the reactants changed over a period of 5 s. She writes in her lab book that the reaction rate is 2.00 10 2...
-
Pizza bought a used Toyota delivery van on January 2, 2016, for $19,200. The van was expected to remain in service for four years (71,200 miles). At the end of its useful life, Piccadilly officials...
-
Explain the importance(s) of the Teamwork soft skill in health care. Describe in detail an example of how it may be used in a healthcare setting.
-
Write a solution to this problem in the main method of a class named " Money " Ask the user to enter a number representing an amount of money from 1 dollar to 9999 dollars (integer). Assume the user...
-
Use linspace to define -4
-
Although beer may be the beverage of choice for most 20 somethings, for 28-year-old Geoff Dillon, his drink of choice would likely be whisky. Dillon grew up watching his dad, an environmental chemist...
-
In our text, the author discusses five drivers of a green supply chain. While each is important, different companies may be more influenced by some more than others. In your discussion, give an...
-
Let and (a) Show that A is positive definite and that xTAx = xTBx for all x R2. (b) Show that B is positive definite, but B2 is not positive definite. A =
-
A fast-food restaurant averages 150 customers per hour. The average processing time per customer is 90 seconds. a. Determine how many cash registers the restaurant should have if it wishes to...
-
Two rocks are thrown off a cliff. One rock (1) is thrown horizontally with a speed of 20 m/s. The other rock (2) is thrown at an angle relative to the horizontal with a speed of 30 m/s. While the...
-
A ball is thrown straight up and rises to a maximum height of 24 m. At what height is the speed of the ball equal to half its initial value? Assume the ball starts at a height of 2.0 m above the...
-
Consider the problem of kicking a soccer ball past a goalkeeper into the goal (Fig. P4.29). You are 25 m away from the goal and kick the ball at an angle of 30° with respect to the horizontal,...
-
Docs Auto Body has budgeted the costs of the following repair time and parts activities for 2009: Doc's budgets 6,000 hours of repair time in 2009. A profit margin of $7 per labour hour will be added...
-
QUESTION 28 In a perpetual inventory system, the cost of inventory sold is: Debited to accounts receivable. Debited to cost of goods sold. O Not recorded at the time goods are sold. O Credited to...
-
The following financial statements and additional information are reported. IKIBAN INC. Comparative Balance Sheets June 30, 2019 and 2018 2019 2018 $105,709 69,500 66,800 4,700 246,700 127,eee...

Study smarter with the SolutionInn App


