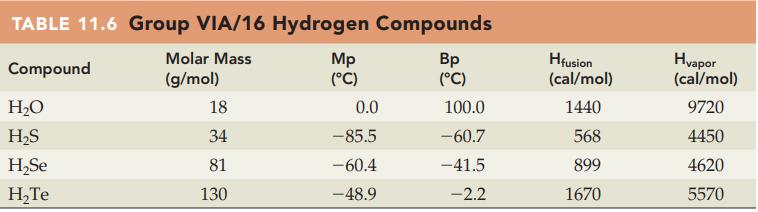
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher
Question:
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher boiling point:
(a) H2O or H2Se
(b) H2S or H2Te.
Table 11.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:





