The tranquilizing drug meprobamate (Equanil or Miltown) can be synthesized from 2-methylpentanal as follows. Give structures for
Question:
The tranquilizing drug meprobamate (Equanil or Miltown) can be synthesized from 2-methylpentanal as follows. Give structures for meprobamate and for the intermediates A–C:
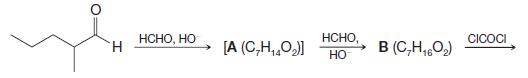
![]()
Transcribed Image Text:
H HCHO, HO [A (C,H,O,)] HCHO. HO в (C,H1602) CICOCI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (9 reviews)
Meprobamate 2methylpentanal In the synthesis of meprobamate A should be given in the name ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Question 20 The relationship between services and providers is much stronger than that between products and producers. True False
-
The tranquilizing drug meprobamate has the structure shown.
-
The tranquilizing drug meprobamate has the structure shown. CH,CH2CH2 CH2OCNH2 HC CHOCNH Meprobamate ROCNH
-
Football Comm LLC is a manufacturer of devices for football coaches and their coaching staffs that enable them to quickly and effectively communicate with one another during a game through a wireless...
-
Let c be the mean number of calories that a person consumes daily, and let w be the persons mean weight (in pounds).
-
Explain four reasons why the quality of data is poor in many organizations.
-
Use the PewKids data to run a logistic regression equation to see if being bullied online is affected by the other three types of bullying: in person, by phone, and by text message. Create a table of...
-
At the end of 2012, Henderson Co. has accounts receivable of $700,000 and an allowance for doubtful accounts of $54,000. On January 24, 2013, the company learns that its receivable from Jaime Lynn is...
-
On January 1, 20 20 , Purat Corporation acquired 75,000 shares of $10 par value common stock out of the 100,000-outstanding stock of Super Corporation. Purat common stock at January 1, 20 20 was...
-
A furnace with an aperture of 20-mm diameter and emissive power of 3.72 x 10 5 W/m 2 is used to calibrate a heat flux gage having a sensitive area of 1.6 x 10 -5 m 2 . (a) At what distance, measured...
-
In general, intramolecular reactions that form rings are often disfavored entropically because it makes a flexible starting material more rigid. In the case of ring-closing metathesis, however, the...
-
Predict the product from each of the following reactions. (a) (b) (c) Br + B(OH) Pd catalyst
-
Refer to Exercise 6.52. Suppose that three people are selected at random. a. Draw a probability tree to depict the experiment. b. If we use the notation RRR to describe the selection of three...
-
A retail product has the following standard costs established: Direct Material per unit - 2 pounds at $5 a pound Direct Labor per unit - 3 hours at $12 an hour Manufacturing Overhead - $5 per labor...
-
In a recent year, the Better Business Bureau settled 75% of complaints they received. (Source: USA Today, March 2, 2009) You have been hired by the Bureau to investigate complaints this year...
-
A 1200-ft equal tangent crest vertical curve is currently designed for 50 mph. A civil engineering student contends that 60 mph is safe in a van because of the higher driver's eye height. If all...
-
Required information [The following information applies to the questions displayed below.] Victory Company uses weighted-average process costing to account for its production costs. Conversion cost...
-
Finer, % 100 90 80 70 60 50 40 30 20 10 0 0.01 0.1 1 Size, mm L 10 100 Figure shows a grain size distribution curve of soil. Estimate the coefficient of curvature (Cc) of this soil.
-
HomeSafe, an accrual basis, calendar year corporation, sells and installs home alarm systems. The contract price of a system includes four free service calls. HomeSafes cost of each call is $75. At...
-
Construct a 4 x 25 design confounded in two blocks of 16 observations each. Outline the analysis of variance for this design.
-
Explain which of the three products shown in is formed when 1-butene reacts with HCI.
-
Explain which of the four products shown in is formed when cis-2-pentene reacts with CI2 andwater.
-
Explain which of the four products shown in is formed when cyclopentene reacts with CI2 andwater.
-
Just work out the assignment on your own sheet, you dont need the excel worksheet. Classic Coffee Company Best friends, Nathan and Cody, decided to start their own business which would bring great...
-
Financial information related to the proprietorship of Ebony Interiors for February and March 2019 is as follows: February 29, 2019 March 31, 2019 Accounts payable $310,000 $400,000 Accounts...
-
(b) The directors of Maureen Company are considering two mutually exclusive investment projects. Both projects concern the purchase of a new plant. The following data are available for each project...

Study smarter with the SolutionInn App


