Predict the major products of the following reactions. a. b. c. d. e. f. g. h. i.
Question:
Predict the major products of the following reactions.
a.
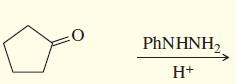
b.
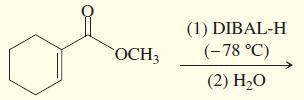
c.
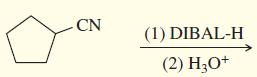
d.

e.
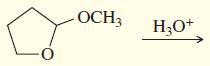
f.
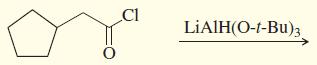
g.

h.
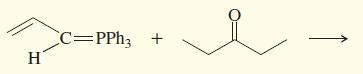
i.
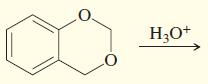
j.
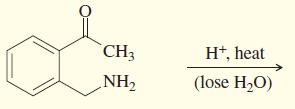
Transcribed Image Text:
PHNHNH2 H+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
c d e f g h i j a b NHNHPh OCH3 PhNHNH H ...View the full answer

Answered By

Ujjwal Ghosh
I have been teaching for last 10 years. Many students whom I've taught are now studying the courses of their choice. Most of my students cracked the competitive examinations in the first attempt.
I've taught many students from graduation to higher level like master degree courses.
They were very happy with my teaching style.
I can solve higher order thinking questions in very less time.
https://www.linkedin.com/in/ujjwal-ghosh-1a506a1b4/
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) CN A OMe O+ OMe A (2) NaBHs (3) H2O OMe MeO 0 CN CN
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Sketch the graph of the function. y = e -x/2
-
Calculate [H+] and pH for the following solutions: (a) 0.010 MHNO3 (b) 0.035 MKOH (c) 0.030 M HCl (d) 3.0 M HCl (e) 0.010 M [(CH3)4N+OH- Tetramethylammonium hydroxide
-
APT Assume that the returns on individual securities are generated by the following two-factor model: R it 5 E ( R it ) 1 b ij F 1 t 1 b i 2 F 2 t Here: R it is the return on Security i at Time t. F...
-
A study of how fast a virus would spread in a metropolitan area
-
The October 31 bank statement of Wollastons Healthcare has just arrived from State Bank. To prepare the bank reconciliation, you gather the following data: a. The October 31 bank balance is $5,580....
-
An investment of $4990.46 earns interest at 42% per annum compounded quarlenly for 4 years Ar fiat frine the Question list quad K inferest rate is changed to 2.5% compounded semi-arinually How much...
-
Julia Walters owns and operates one of the largest Mercedes-Benz auto dealerships in Washington, DC. In the past 36 months her sales of this luxury car have ranged from a low of 6 new cars to a high...
-
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name. a. CH 3 CO(CH 2 )4CH 3 b. CH 3 (CH 2 ) 2 CO(CH 2 ) 2 CH 3 c. CH 3 (CH 2 ) 5 CHO d. PhCOPh e. CH 3...
-
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration: CH,COCH,CI CICH,CHO CH,0 CH3COCH3 CH3CHO
-
A pump for a reservoir must be operated continuously (8,760 hours per year). In the event of a large storm, the electricity from the local utilitys power grid may be interrupted for an indefinite...
-
Consider a piston with an orifice in a cylinder filled with a fluid of viscosity \(\mu\) as shown in Fig. 1.106. As the piston moves in the cylinder, the fluid flows through the orifice, giving rise...
-
Add a function to SmallWorld that computes the global clustering coefficient of a graph. The global clustering coefficient is the conditional probability that two random vertices that are neighbors...
-
Show that the generators of the algebra (33.8) are related by parity. For a Dirac wavefunction the action of parity is $P \psi(\boldsymbol{x}, t) P^{-1}=\gamma_{0} \psi(-\boldsymbol{x}, t)$, up to a...
-
Extend the algorithm you designed for Exercise 6.2 so that it can evaluate positions that are nonterminalin other words, positions where the game has not yet finished. Your score should be positive...
-
In addition to tanh, another s-shaped smooth function, the logistic sigmoid function y=1 / (1+exp(x)), is commonly used as an activation function in neural networks. A common way to implement them in...
-
The two attached pulleys are driven by the belt with increasing speed. When the belt reaches a speed v = 2 ft/sec, the total acceleration of point P is 26 ft/sec 2 . For this instant determine the...
-
The figure shows a bolted lap joint that uses SAE grade 8 bolts. The members are made of cold-drawn AISI 1040 steel. Find the safe tensile shear load F that can be applied to this connection if the...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices. (1) (2)
-
ABC Corporation has an activity - based costing system with three activity cost pools - Machining, Setting Up , and Other. The company's overhead costs, which consist of equipment depreciation and...
-
Consolidated Balance Sheets - USD ( $ ) $ in Thousands Dec. 3 1 , 2 0 2 3 Dec. 3 1 , 2 0 2 2 Current assets: Cash and cash equivalents $ 9 8 , 5 0 0 $ 6 3 , 7 6 9 Restricted cash 2 , 5 3 2 Short -...
-
How does corporate governance contribute to investor confidence and stakeholder trust? Accounting

Study smarter with the SolutionInn App


