Classify each of the labeled bonds in the following structure in terms of the bond type (
Question:
Classify each of the labeled bonds in the following structure in terms of the bond type (σ or π) and the component orbitals that overlap to form the bond. (For example, the carbon–carbon bond in ethane is an sp3–sp3 s bond.)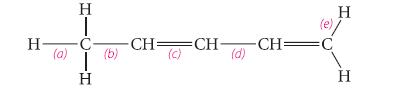
Transcribed Image Text:
H- (a) HI T H (b) -CH=CH- (c) (d) (e) -CH=C H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a b c d e An sp C1sH or ...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Classify each of the labeled bonds in the following structure in terms of the bond type ( or ) and the component orbitals that overlap to form the bond. (For example, the carbon--carbon bond in...
-
For each of the following compounds and ions, 1. Draw a Lewis structure. 2. Show the kinds of orbitals that overlap to form each bond. 3. Give approximate bond angles around each atom except...
-
Draw the two resonance structures that describe the bonding in the acetate ion. What is the hybridization of the carbon atom of the CO 2 group? Select one of the two resonance structures and...
-
M DO NOT TURN THIS PAGE UNTIL YOU ARE TOLD TO DO SO SECTION B. ANSWER ANY THREE (3) QUESTIONS Question 1 (25 marks) You have analyzed Solar Power Ltd. and have reported the results of your analysis...
-
Some states make liquor stores, bars, and restaurants liable for serving drinks to intoxicated persons (minors or adults) who later cause harm, while not holding social hosts liable. Why the...
-
Consider a system consisting of two oppositely charged spheres hanging by strings and separated by a distance r1, as shown in the accompanying illustration. Suppose they are separated to a larger...
-
A sample of automobile dealerships found that 19% of automobiles sold are silver, 22% of automobiles sold are sport utility vehicles (SUVs), and 16% of automobiles sold are silver SUVs. What is the...
-
In its response to problems in its contractor factories, do you think Apple moved through the stages of corporate citizenship presented in this chapter? Why do you think so?
-
Wail Inc. is currently a firm that has 2 million shares of stock outstanding with a market price of $25 a share and outstanding debt of $30 million. The debt interest rate is 10%. Its cost of equity...
-
A confused chemist Al Keane used the following names in a paper about alkenes. Although each name specifies a structure, in some cases the name is incorrect. Correct the names that are wrong. (a)...
-
Give a structure for each of the following compounds. (a) Cyclobutene (b) 3-methyl-1-octene (c) 5,5-dimethyl-1,3-cycloheptadiene (d) 1-vinylcyclohexene
-
Calculate the minimum possible magnitude \(u\) of the muzzle velocity which a projectile must have when fired from point \(A\) to reach a target \(B\) on the same horizontal plane \(12 \mathrm{~km}\)...
-
You are the cost accountant of an engineering concern which has three departments - preparation, machining and assembly. The budgeted direct labour hours for the workshops are 8,000, 12,000 and...
-
What alternative to fostering fun and enjoyment at work do you think might have worked for Zappos?
-
Using the techniques of dimensional analysis, and assuming that experimentation shows the dimensionless number to be 1, derive the following equation: E v = Job card two The results of an ultrasonic...
-
Given the historical cost of product Carla Vista is $13, the selling price of product Carla Vista is $15, costs to sell product Carla Vista are $3, the replacement cost for product Carla Vista is...
-
What causes of outliers in statistics and when I create a boxplot why do I not see the outliers. What steps are to take in creating a boxplot?
-
Sometimes, in response to irritants, mucous cells secrete excess mucus. What symptoms might this produce if it occurred in the (a) Digestive tract or (b) Respiratory passageway?
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities. (a) 2-Methyl-2-propanol (b) 1-Ethylcyclohexanol (c) 3-Phenyl-3-pentanol...
-
Evidence for the intermediate carbocations in the acid-catalyzed dehydration of alcohols comes from the observation that rearrangements sometimes occur. Propose a mechanism to account for the...
-
Read the following and then answer the questions below:September 12: A Brisbane business offers by letter to sell 500 tyres to a New Zealand company. The Brisbane company does not specify a method of...
-
Fred returns home from work one day to discover his house surrounded by police. His wife is being held hostage and threatened by her captor. Fred pleads with the police to rescue her and offers...
-
Would like you to revisit one of these. Consideration must be clear and measurable.if you can't measure it then how can you show it has / has not been done?How can you sue someone for breach of...

Study smarter with the SolutionInn App


