For each of the following balanced oxidationreduction reactions, indicate which compound(s) are oxidized and which are reduced.
Question:
For each of the following balanced oxidation–reduction reactions, indicate which compound(s) are oxidized and which are reduced.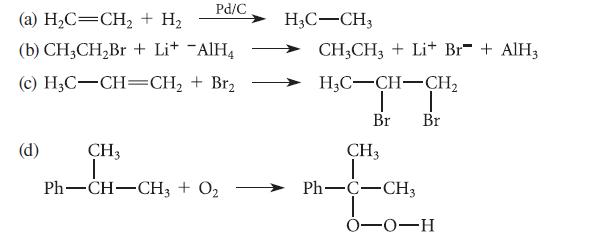
Transcribed Image Text:
Pd/C (a) H₂C=CH₂ + H₂ (b) CH3CH₂Br + Li+ AlH4 (c) H3C-CH=CH₂ + Br₂ (d) CH3 T Ph–CH–CH3 + O2 H3C-CH3 CH3CH3 +Li+ Br+AlH3 H3C-CH-CH₂ I T Br Br CH3 Ph—C–CH3 0-0-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a The alkene is reduced and the H is oxidi...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
KitchenPaid (KP) is a U.S. manufacturer of upscale small kitchen appliances. As a result of an acquisition made in 2013 KP also has a small division that manufactures medical supplies. KP is...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) CH3C = PCH +...
-
Doug, Peter, and Jack have the following capital balances;$150,000, $300,000 and $320,000, respectively. The partners shareprofits and losses 35%, 40%, and 25% respectively. Jones is goingto pay a 2...
-
Refer to the data in PE 6-16. Calculate the companys average collection period. Data from PE 6-16 Accounts receivable balance, December 31 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Before starting this problem, review Conceptual Example 3. Suppose that the hail described there bounces off the roof of the car with a velocity of + 15 m/s. Ignoring the weight of the hailstones,...
-
Why would the members of a NOT work independently if they were members of a designated team? What does independently mean in this context? LO6
-
A gold-mining firm is concerned about short-term volatility in its revenues. Gold currently sells for $300 an ounce, but the price is extremely volatile and could fall as low as $280 or rise as high...
-
Suppose the current exchange rate for the Polish zloty is Z 2.84. The expected exchange rate in three years is Z 2.92. What is the difference in the annual inflation rates for the United States and...
-
Contrast the products expected when 2-methyl-3-pentanol is treated with (a) HBr/H 2 SO 4 or (b) Ph 3 PBr 2 . Explain.
-
Give the structure of two secondary alcohols that could be converted by HBr/H 2 SO 4 into the corresponding alkyl bromide without rearrangement.
-
What implications for managerial spans of control can be expected as organizations downsize? What additional managerial competencies might be required of leaders who remain in a downsized...
-
I need help with discussion posts that respond to 3 of these comments. 2 of them being the first on each picture. RUBRIC: articles to mention Coleman, R., & Banning, S. (2006). Network TV news'...
-
2. Best Use of Scarce Resource DigiCom Corporation produces three sizes of television sets: 12-inch screen, 26-inch screen, and 40-inch screen. Revenue and cost information per unit for each product...
-
Gunther invested $15,000 into a segregated fund with a 65% maturity guarantee 10 years ago. The fund is now maturing and has a current market value of $22,261. Gunther decides to withdraw his...
-
(a) Consider the following financial data (in millions of dollars) for Costello Laboratories over the period of 2014-2018: Year Sales Net income Total assets Common equity 2014 $3,800 $500 $3,900...
-
The Pizza Pie 'N Go sells about 2300 one-topping pizzas each month. The circle graph displays the most requested one-topping pizzas, by percentage, for one month. Most Popular One-Topping Pizzas...
-
Sketch the graph of G(t) = t - [t].
-
Evaluate the line integral, where C is the given curve. C x 2 dx + y 2 dy, C consists of the arc of the circle x 2 + y 2 = 4 from (2, 0) to (0, 2) followed by the line segment from (0, 2) to (4, 3)
-
Write the equation for the final step in the -oxidation pathway of any fatty acid with an even number of carbon atoms.
-
Show the products of each of the followingreactions: (a) FAD FADH2 CHCH-CH2CH2CHzCsCOA Acyl-CoA dehydrogenase (b) Enoyl-CoA hydratase Product of (a) + H20 NAD+ NADH/H* (c) Product of (b)...
-
What is the structure of the a-keto acid formed by transamination of each of the following amino acids? (a) Threonine (b) Phenylalanine (c) Asparagine
-
The star Mira is 1.2 times the mass of the Sun and about 10,000 times more luminous than the Sun. Would Mira fit into the table above? Why or why not?
-
Which of the following was one of the most valuable benefits a company received as a sponsor of NHL games?
-
Cinder Inc. is a Canadian-controlled private corporation based in your province. The company operates a wholesale business. The following information is provided for its year ended May 31, 2023: Net...

Study smarter with the SolutionInn App


