Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in
Question:
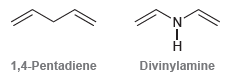
The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using this information, identify the hybridization state of the nitrogen atom in divinylamine, and justify your answer.
'N' Divinylamine 1,4-Pentadiene
Step by Step Answer:
The nitrogen atom in divinyl amine is sp 2 hybridized The lone pai...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
Compare the structures of a synchondrosis and a symphysis.
-
Compare the structures of lanosterol and cholesterol, and catalog the changes needed for the transformation.
-
Compare the structures of HNO2 and H2CO3. Which would you expect to be the stronger acid? Explain your choice.
-
1. The Jarmon Company manufactures and sells a line of exclusive sportswear. Prepare a financial position as at 31st December 2022 from the following information. Account Receivable Long term loan...
-
A sample of sand has a relative density of 40% with a specific gravity of solids of 2.65. The minimum void ratio is 0.45 and the maximum void ratio is 0.97. (a) What is the unit weight (in units of...
-
The diameter of the sun is 1.38 10 6 km, the diameter of the Earth is 1.27 10 4 km, and the distance from the Earth to the sun (center to center) is 1.50 10 8 km. What is the distance from the center...
-
Im like the quarterback of the team. I manage 250 accounts, and anything from billing issues, to service issues, to selling the products. Im really the face to the customer, says Alison Capossela, a...
-
Prior to adjustment at the end of the year, the balance in Trucks is $275,900 and the balance in Accumulated Depreciation'Trucks is $91,350. Details of the subsidiary ledger are as follows: a....
-
Exercise 11-11A (Algo) Fixed versus variable cost behavior LO 11-1, 11-2 Solomon Entertainment sells souvenir T-shirts at each rock concert that it sponsors. The shirts cost $10 each. Any excess...
-
Comparative balance sheets for 2024 and 2023 and a statement of income for 2024 are given below for Metagrobolize Industries. Additional information from the accounting records of Metagrobolize also...
-
How do perspectives on climate change of highly developed countries differ from those of less developed countries?
-
Provide a systematic name for each of the following compounds. a. b. c. d. e. H. CH3 Br
-
Compare and contrast incremental and zero-based budgeting.
-
1. Write short notes on Discrete Cosine Transform (DCT) 2. Describe Fast Fourier Transform 3. Write short notes on the Hotelling transform
-
1Explain Discrete Fourier Transform in detail. 2. Explain the Properties of 2D discrete Fourier Transform 3. Discuss Hadamard's transformation in detail
-
1. Specify the objective of image enhancement technique. 2. List the 2 categories of image enhancement. 3. What is the purpose of image averaging? 4. What is meant by masking? 5. Define histogram.
-
6. What is meant by histogram equalization? 7. Differentiate linear spatial filter and non-linear spatial filter. 8. Give the mask used for high boost filtering. 9. What is meant by a laplacian...
-
11. What do you mean by Point processing? 12. Define Derivative filter? 13. Define spatial filtering 14. What is a Median filter? 15. What is maximum filter and minimum filter? 16. Write the...
-
Find each product. (m 5p)(m 2mp + 3p)
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2 "CH2 - H Hydroxyacetone Crotyl alcohol
-
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the Re face, what is the stereochemistry of theproduct? OH CH3CHCO2 H3C CO2 Lactate Pyruvate
-
The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or the Si face of the...
-
In 2019, Sunland Company had a break-even point of $388,000 based on a selling price of $5 per unit and fixed costs of $155,200. In 2020, the selling price and the variable costs per unit did not...
-
11. String Conversion Given a binary string consisting of characters '0's and '1', the following operation can be performed it: Choose two adjacent characters, and replace both the characters with...
-
Consider the table shown below to answer the question posed in part a. Parts b and c are independent of the given table. Callaway Golf (ELY) Alaska Air Group (ALK) Yum! Brands (YUM) Caterpillar...

Study smarter with the SolutionInn App


