Consider the structures of cis-decalin and trans-decalin: (a) Which of these compounds would you expect to be
Question:
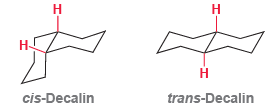
(a) Which of these compounds would you expect to be more stable?
(b) One of these two compounds is incapable of ring flipping. Identify it and explain your choice.
Transcribed Image Text:
Н cis-Decalin trans-Decalin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a CisDecalin has three gauche interactions while transdecalin has o...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of these compounds would you expect to be more soluble in water? Explain? CHCHCHCHCOH or CH3CHCHCHCHCOH
-
Consider the structures of cis-1,2-dimethylcyclopropane and trans-1,2-dimethylcyclopropane: (a) Which compound would you expect to be more stable? Explain your choice. (b) Predict the difference in...
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
On 28 April 2020, Mr Guna, CEO of Econ Engineering Malaysia, proposed to complete an abandoned boiler project that no one had dared to revive. He knew that the project was 60% complete before it was...
-
The Bretton Woods institutions were created in the aftermath of World War II. Many believe they are obsolete or are in need of massive redesign. The way forward must reference the road that has been...
-
Prove that the continuoustime LTI system (15.20) is asymptotically stable (or stable, for short) if and only if all the eigenvalues of the A matrix, /(A), i = 1, . . . , n, have (strictly) negative...
-
Apply the quality concepts, methods, and tools introduced in this chapter to develop a project quality plan. AppendixLO1
-
When is a companys competitive advantage most likely to endure over time?
-
Doaktown Products manufactures fishing equipment for recreational uses. The Miramichi plant produces the companys two versions of a special reel used for river fishing. The two models are the M-008,...
-
Here is the case: (Marian could feel the rage surge from deep within her. Even though she was usually in control of her behavior, it was not easy to control her internal emotions. She could sense...
-
A housing bubble occurs when _________ drive(s) prices more than fundamental factors. a. The price of gasoline b. A homes expected future price c. Interest rate changes d. Property tax increases
-
Atorvastatin is sold under the trade name Lipitor and is used for lowering cholesterol. Annual global sales of this compound exceed $13 billion. Assign a configuration to each chirality center in...
-
As you increase the length of time involved, what happens to future values? What happens to present values?
-
How would you explain the following code in plain English? boxplot(age ~ gender, data = donors) Question 8 options: Make a boxplot comparing gender grouped by age, using the donors dataset Make two...
-
Vision Consulting Inc. began operations on January 1, 2019. Its adjusted trial balance at December 31, 2020 and 2021 is shown below. Other information regarding Vision Consulting Inc. and its...
-
A Jeans maker is designing a new line of jeans called Slams. Slams will sell for $290 per unit and cost $182.70 per unit In variable costs to make. Fixed costs total $68,500. (Round your answers to 2...
-
NAME: Week Two Define Claim in your own words Explain the difference between a discussion and an argument. Summarize the characteristics of a claim (Listing is not summarizing) Define Status Quo in...
-
1.How do you think major stores such as Walmart will change in the future under this new retail renaissance? 2.What are some changes that you would suggest in traditional retail stores to attract...
-
In Exercises 3744, find the horizontal asymptote, if there is one, of the graph of each rational function. f(x) = 12x 3x + 1
-
The percentage of completion and completed contract methods are described in the FASB ASC. Search the codification to find the paragraphs covering these topics, cite them, and copy the results.
-
Suggest explanations for the origins of "ibu," "pro," and "fen" in the name ibuprofen. Provide a systematic name for thiscompound OH O,N. NO2 NO2 Picric acid
-
The pK a for the picric acid is 0.42. Explain why it is such a strong acid.
-
To find a base that is strong enough to deprotonate benzoic acid but not p-methyl phenol. Then explain how this base might be used to separate these two compounds in the laboratory.
-
In 2019, Sunland Company had a break-even point of $388,000 based on a selling price of $5 per unit and fixed costs of $155,200. In 2020, the selling price and the variable costs per unit did not...
-
11. String Conversion Given a binary string consisting of characters '0's and '1', the following operation can be performed it: Choose two adjacent characters, and replace both the characters with...
-
Consider the table shown below to answer the question posed in part a. Parts b and c are independent of the given table. Callaway Golf (ELY) Alaska Air Group (ALK) Yum! Brands (YUM) Caterpillar...

Study smarter with the SolutionInn App


