Determine whether the following compounds are constitutional isomers: CH - H3C H;C CH3 CH3
Question:
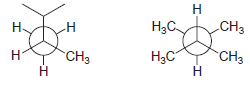
Transcribed Image Text:
Н CHз -Н H3C Н H;C CH3 CH3 Н Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
The compounds are not consti...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether the following compounds are soluble or insoluble in water. a. NaBr b. Ba(OH)2 c. Calcium carbonate
-
Consider the three compounds shown below and then answer the questions that follow: a) Which two compounds are constitutional isomers? b) Which compound contains a nitrogen atom with trigonal...
-
For each of the following pairs of compounds, identify whether the compounds are constitutional isomers or different representations of the same compound: (a) (b) (c)
-
A health club has cost and revenue functions given by C = 10,000 + 35q and R = pq, where q is the number of annual club members and p is the price of a one year membership. The demand function for...
-
There are three questions related to the XML technologies and history. Use the following website: www.w3schools.com /xm Please answer the three research questions: 1. How/why was XML created and how...
-
Let A be an n n matrix and let B = A + AT and C = A - AT (a) Show that B is symmetric and C is skew-symmetric. (b) Show that every n n matrix can be represented as a sum of a symmetric matrix and a...
-
Identify the services offered by the different financial institutions.
-
Mile-High Foods, Inc., was formed in March 2011 to provide prepackaged snack boxes for a new low cost regional airline beginning on April 1. The company has just leased warehouse space central to the...
-
. Problem 13-15 Capital Structure (LO3) 10 In 2020, Caterpillar Incorporated had about 540 million shares outstanding. Their book value was $25.36 per share, and the market points price was $153.70...
-
The following information was taken from the financial statements of Tolbert Inc. for December 31 of the current fiscal year: Common stock , $20 par (no change during the year).... $10,000,000...
-
Draw a bond-line structure for each of the following compounds: (a) (b) (c) CH CH- CH2CH3 CH CH-CH CH . CH
-
For each of the following compounds, predict the energy barrier to rotation (looking down any one of the C-C bonds). Draw a Newman projection and then compare the staggered and eclipsed...
-
How has the Fed used mortgage-backed securities in recent years, and what has it been trying to accomplish?
-
Sierra Company manufactures soccer balls in two sequential processes: Cutting and Stitching. All direct materials enter production at the beginning of the cutting process. The following information...
-
Q5A. Evaluate the arithmetic series 222n + 7. -220
-
The football team is planning a trip to a sports museum. The cost for renting a bus is $ 3 6 0 . The cost will be divided equally among those going on the trip. A museum ticket costs $ 1 4 . 2 5 per...
-
Factor by grouping. 8x+8y+ax+ay
-
BuyCo, Incorporated, holds 2 5 percent of the outstanding shares of Marqueen company and appropriately applies the equity method of accounting. Excess cost amortization ( related to a patent )...
-
A quick quiz consists of a true/false question followed by a multiple-choice question with four possible answers (a, b, c, d). An unprepared student makes random guesses for both answers. a. Consider...
-
Show that every group G with identity e and such that x * x = e for all x G is abelian.
-
The rings below are joined by a double bond that undergoes cis-trans isomerization much more readily than the bond of a typical alkene. Provide an explanation.
-
Although Hückel's rule (Section 14.7) strictly applies only to monocyclic compounds, it does appear to have application to certain bicyclic compounds, if one assumes use of resonance...
-
(a) In 1960 T. Katz (Columbia University) showed that cyclooctatetraene adds two electrons when treated with potassium metal and forms a stable, planar dianion, C8H82- (as the dipotassium salt): Use...
-
Complete Programming Exercise 3.17 found at the end of Chapter 3. Call this program Game.java. 1. Use the Random Class and the nextInt() method to generate random numbers in the range of 0 - 2. 2....
-
Caren's Creamy Creations recruited a managerial accountant to help forecast the company's expenses and income for its current year in business. The plan for how the baking company plans to grow from...
-
2 D Bohimil has a T4 slip that indicates a pension adjustment of $8,900. What statement is true? O a) Bohimil must include this amount in his taxable income. Ob) Bohimil can deduct this amount from...

Study smarter with the SolutionInn App


