Draw the mechanism of the following reaction, and make sure to draw all three resonance structures of
Question:
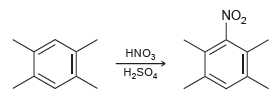
Transcribed Image Text:
NO, HNO3 H,SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
28 H oja OTH H ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the mechanism of the following reaction. This reaction is the reverse of sulfonation, so you should read the sulfonation mechanism backward. Your mechanism should involve a sigma complex...
-
Draw the mechanism of the following reaction, which involves two consecutive Friedel-Crafts alkylations. When drawing the mechanism, do not try to draw the two alkylations as occurring simultaneously...
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
Karamazov Semiconductors is considering an investment to expand its existing line of business. The investment will cost $10 million and is expected to produce after-tax cash flows of $1 million per...
-
Visit the Web site of the National Institute of Standards and Technology (www.nist.gov/baldrige/), the organization that grants the Malcolm Baldrige National Quality Award, the highest quality award...
-
Describe the magic triangle.
-
Picture Gallery, Inc. works weddings and prom-type parties. The balance of retained earnings was \($18,000\) at December 31, 2010. At December 31, 2011, the businesss accounting records show these...
-
Change in Accounting Principle Zehms Company began operations in 2008 and adopted weighted-average pricing for inventory. In 2010, in accordance with other companies in its industry. Zehms changed...
-
The local school district was planning to build a new high school. The estimated cost of the building was around $100M. To finance the building the school district needed to issue a bond for about...
-
The following balance sheet, which has some weaknesses in terminology and classification, has been prepared by an inexperienced accountant and submitted to you for review: Required: Prepare a...
-
Calculate KP at 298 and 490. K for the reaction NO(g) + 1/2O 2 (g) NO 2 (g) assuming that H o R is constant over the interval 298600. K. Do you expect K p to increase or decrease as the temperature...
-
A sample containing 2.75 moles of N 2 and 6.25 mol of H 2 is placed in a reaction vessel and brought to equilibrium at 52.0 bar and 690. K in the reaction 1/2N 2 (g) + 3/2H 2 (g) NH 3 (g). a....
-
An ethanol-water vapor mixture is being rectified by contact with an alcohol-water liquid solution. Alcohol is being transferred from gas to liquid and water from liquid to gas. The molal flow rates...
-
Capsule endoscopy is used to examine parts of the gastrointestinal tract that cannot be seen with other types of endoscopy. The process uses a very small camera attached to a long flexible tube to...
-
In March 2015, a Nielsen global online survey "found that consumers are increasingly willing to pay more for socially responsible products." Over 30,000 people in 60 countries were polled about their...
-
A long-term care facility (50 beds) in the Midwestern U.S. has just been acquired by a larger acute care hospital/health care system in the nearby city because they want to offer services across the...
-
Annie is the manager of a Health and Wellness Center at Tippy Canoe University Medical Center (TCUMC), a facility that has been known regionally in the mid-west for its research and cutting-edge...
-
Correlation between height and weight for players on the 2014 Brazil World Cup Team, using data from all 23 players on the roster. State whether the quantity described is a parameter or a statistic...
-
The principle that receipts should be banked daily and all payments made by cheque or electronic transfer provides a sound basis for internal control over cash. But it breaks down immediately when...
-
Pearl Medavoy will invest $10,240 a year for 20 years in a fund that will earn 10% annual interest. . If the first payment into the fund occurs today, what amount will be in the fund in 20 years? If...
-
In a laboratory, two liquids, A and B, were found in a box labeled only "isomeric alkyl halides C5H11Br." You have been employed to deduce the structures of these compounds from the following data...
-
What is the difference in the standard free energies of activation at 25oC of two reactions A and B if reaction B is 450 times faster than reaction A ? Which reaction has the greater G?
-
Which one of the following stereoisomers should undergo β-elimination most rapidly with sodium ethoxide in ethanol? Explain your reasoning. CH1 CH3 Br CH3 CH
-
EXERCISE 3-2 (LO1,2) Adjusting Entries The trial balance of Lauer Corporation at December 31, 2015 follows, before and after the posting of adjusting entries. Exercises 131 Trial Balance Adjustments...
-
Question 4 Let f(z) = 1 1 - a cos a-1 x+|x| x3+27 if x-3 if x = -3 Suppose f is continuous at every point on the whole real line R. Determine the value of a.
-
Write a new warehouse health and safety policy that defines how issues must be reported to management.

Study smarter with the SolutionInn App


