Identify the reagents necessary to accomplish each of the following transformations. >
Question:
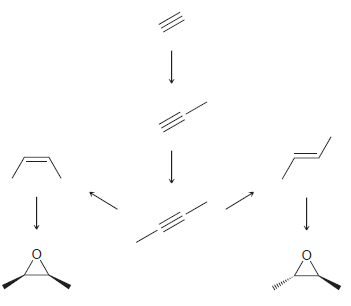
Transcribed Image Text:
>
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
MCPBA 1 NaNH ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b) Me
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b) OH HO NH2
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b)
-
Three employees of the Horizon Distributing Company will receive annual pension payments from the company when they retire. The employees will receive their annual payments for as long as they live....
-
On January 1, 2010, ABC Co. had inventory of 200 units @ 18.00 a unit. It purchased 400 more units @ $20.00 a piece on Feb. 19th. On May 12th, it bought 300 more units @ $22.00 each. On Oct. 3rd, it...
-
Who should be an entrepreneur and who should not?
-
What is the purpose of performance measurement? LO.1
-
Mary Janus is developing a transfer price for the housing section of an automatic pool-cleaning device. The housing for the device is made in Department A. It is then passed on to Department D, where...
-
An earnings and profits (E&P) study for Hardy Corp., a calendar year C corporation was performed. The results of the study indicated that Hardy had accumulated E&P of $50,000 as of January 1 st ,...
-
Given a database of the results of an election, find the number of seats won by each party. There are some rules to going about this: There are many constituencies in a state and many candidates who...
-
Methylmagnesium bromide reacts rapidly with ethylene oxide, it reacts slowly with oxetane, and it does not react at all with tetrahydrofuran. Explain this difference in reactivity. Oxetane Ethylene...
-
When 5-bromo-2, 2-dimethyl-1-pentanol is treated with sodium hydride, a compound with molecular formula C 7 H 14 O is obtained. Identify the structure of this compound. NaH, Br -40
-
Jim Shea is an accountant at King and Associates, which is an accounting firm based in Halifax, Nova Scotia. The firm specializes in dealing with small business clients who generally are very...
-
1. A corn farmer has observed the following distribution for the number of ears of corn per cornstalk. Ears of Corn Probability 1 2 3 4 .3 .4 .2 .1 Part A: How many ears of corn does he expect on...
-
1. A mass m on a vertical spring with force constant k has an amplitude of A. Using the top of the motion as the origin for both gravitational potential energy and spring potential energy: (a) Find...
-
2. Consider the PDE Utt - Uxx + Ut - Ux = 0 (1) for < < and 0
-
On April 1, 2024, Chardonnay pays an insurance company $12,480 for a two- year fire insurance policy. The entire $12,480 is debited to Prepaid Insurance at the time of the purchase. Record the...
-
Which retailer(s) should represent and sell your product?Why?In terms of their range of distribution coverage, is your retailer intensive, selective and exclusive? Why is this aspect important to...
-
Explain what the following statement means: The solution set of the following system is {(-1, 2, 3)}. 2x + y + z = 3 3x - y + z = 2 -2 4xy + 2z = 0
-
Give the structural formulas of the alkenes that, on ozonolysis, give: a. (CH3)2C=O and CH2=O b. Only (CH3CH2)2C=O c. CH3CH=O and CH3CH2CH=O d. O=CHCH2CH2CH2CH=O
-
One of the steps in the pentose phosphate pathway for glucose catabolism is the reaction of sedoheptulose 7-phosphate with glyceraldehydes 3-phosphate in the presence of a transaldolase to yield...
-
One of the steps in the pentose phosphate pathway for glucose catabolism is the reaction of xylulose 5-phosphate with ribose 5-phosphate in the presence of a transketolase to give glyceraldehydes...
-
The amino acid tyrosine is biologically degraded by a series of steps that include the following transformations: The double-bond isomerization of maleoylacetoacetate to fumaroyl acetoacetate is...
-
(1 point) Bill makes annual deposits of $1900 to an an IRA earning 5% compounded annually for 14 years. At the end of the 14 years Bil retires. a) What was the value of his IRA at the end of 14...
-
Which of the following concerning short-term financing methods is NOT CORRECT? Short-term bank loans typically do not require assets as collateral. Firms generally have little control over the level...
-
Kingbird Corporation is preparing its December 31, 2017, balance sheet. The following items may be reported as either a current or long-term liability. 1. On December 15, 2017, Kingbird declared a...

Study smarter with the SolutionInn App


