Nitrogen and sulfur are neither in the same row nor in the same column of the periodic
Question:
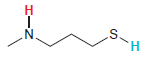
Transcribed Image Text:
.S. H. エーZ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
A proton connected to a sulfur atom will be more acidi...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ammonium sulfate is used as a nitrogen and sulfur fertilizer. It is produced by reacting ammonia with sulfuric acid. Write the balanced equation for the reaction of gaseous ammonia with sulfuric acid...
-
Which of the hydrogen atoms shown below is more acidic? Explain your answer. A
-
The diagram that follows shows the highest-energy occupied MOs of a neutral molecule CX, where element X is in the same row of the periodic table as C. (a) Based on the number of electrons, can you...
-
In Exercises, find the limit. lim (4x- x 16x - x
-
One area that we haven't looked at yet is payroll. Can you get the documentation from Karina for payroll and then complete a "bridge work paper" like you did for the Revenue and Purchasing cycles? As...
-
Starting with methyl propanoate, how could you prepare 4-methyl-3-heptanone? CH3CH3COCH3CH3CH CCHCH CH2CH3 methyl propanoate CH3 4-methyl-3-heptanone
-
Dahlia Garcia is contemplating the purchase of a machine that will automate the production of hosiery in her factory. The following estimates are available: Required: Determine the net present value...
-
Colaw Stores accepts both its own and national credit cards. During the year, the following selected summary transactions occurred. Jan. 15 Made Colaw credit card sales totaling $18,000. (There were...
-
Only need help for F and H-1. Consider the following two projects: Cash flows Project A Project B Co -$150 -$150 C1 60 75 60 75 C3 60 75 CA 60 a. If the opportunity cost of capital is 10%, which of...
-
Pane records depreciation and amortization expense. Use the nearest whole month convention, and the double declining balance method for depreciation and the whole-year convention and the...
-
In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. (a) (b) (c) (d) N' C-
-
In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. (a) (b) (c) (d) (e) (f) -N
-
List the five basic steps of demand forecasting in supply chain management. Which step is more important?
-
1. Discuss the message that's hidden in the financial ratios you collected/calculated. Provide suggestions if possible A: Ratio Analysis for Year 2021 Ratio Current Ratio Quick Ratio Inventory...
-
Texas Innovation (TI) (U.S) is considering investing Rs100,000,000 in India to create a wholly owned tile manufacturing plant. After five years the subsidiary would be sold to Indian investors for...
-
Mulligan Manufacturing Company uses a job order cost system with overhead applied to products at a rate of 150 percent of direct labor cost. Required: Treating each case independently, selected from...
-
The Polaris Company uses a job-order costing system. The following transactions occurred in October: a. Raw materials purchased on account, $209,000 b. Raw materials used in production, $189,000...
-
Determine the heat rejected in kJ / kg to a simple ideal steam Rankine cycle with a condenser pressure of 2 0 kPa and a steam generator with a pressure of 2 MPa. ( The steam leaves the steam...
-
Many nonparametric tests are based on ranks. What is a rank? What are the ranks of the following lengths (in hr) of NASA Space Shuttle Transport System flights (from Data Set 10 in Appendix B): 54,...
-
Which internal control principle is especially diffi cult for small organizations to implement? Why?
-
The halogen atom of an alkyl halide can be replaced by the hydrogen atom bonded to tin in tributyltin hydride (Bu3SnH). The process, called dehalogenation, is a radical reaction, and it can be...
-
Write a mechanism that accounts for the following reaction. The hydrogen atom bonded to tin in tributyltin hydride is readily transferred in radical mechanisms. Br PhCO22, BuSnH (Major)
-
Molecular orbital calculations can be used to model the location of electron density from unpaired electrons in a radical. Open the molecular models on the book's website for the methyl, ethyl, and...
-
Cola Drink Company processes direct materials up to the splitoff point where two products, A and B, are obtained. The following information was collected for the month of July: Direct materials...
-
Required information [The following information applies to the questions displayed below.] Hudson Company reports the following contribution margin income statement. HUDSON COMPANY Contribution...
-
Methods that do not use present value One category of capital investment evaluation methods does not use present value. The primary difference between the category of methods that do use present...

Study smarter with the SolutionInn App


