Predict the major product(s) for each of the following reactions: a. b. c. d. Br2 Br2
Question:
a.
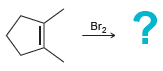
b.
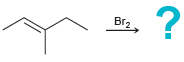
c.
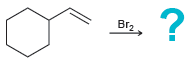
d.
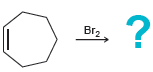
Transcribed Image Text:
Br2 Br2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a b ...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
.How dependent do you think the success of zero-based budgeting would be on the types of attribution models used? How might you increase the odds of success in this budgeting approach? .Advertising,...
-
What authorizations does the AR Supervisor not have for Account Types? Why do you think the AR Supervisor would be restricted from these authorizations for this accounting document?
-
Simplify the given expressions involving the indicated multiplications and divisions. 6x + 5 4x5x6 x2 X 12x + 9 x - 1
-
Deer in the suburbs. Westchester County is a suburban area covering 438 square miles immediately north of New York City. A garden magazine claimed that the county is home to 800,000 deer. Do a...
-
What is interim financial information, and why is interim financial information required to be reviewed? What professional standards provide guidance on the conduct of reviews of interim financial...
-
QUESTION 4 4.1 REQUIRED Calculate the following from the information given below: Payback Period (expressed in years). Return on investment (expressed to two decimal places). INFORMATION Tridev...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
Compound X has molecular formula C 5 H 10 . In the presence of a metal catalyst, compound X reacts with one equivalent of molecular hydrogen to yield 2-methylbutane. a. Suggest three possible...
-
Predict the major product(s) that are expected when each of the following alkenes is treated with Br 2 /H 2 O: a. b. c. d.
-
One way insurance companies reduce adverse selection problems is by offering group medical coverage to large firms and requiring all employees to participate in the coverage. Explain how this reduces...
-
REQUIRED: Cost of production report under the following assumptions: Lost units - normal, discovered at the beginning Lost units - normal, discovered at the end Lost units - abnormal, discovered when...
-
ABC, Inc., manufactures only two products: Gadget A and Gadget B. The firm uses a single, plant wide overhead rate based on direct-labor hours. Production and product-costing data are as follows:...
-
.Jean Saburit has gone over the financial statements for Saburit Parts, Inc. The income statement has been prepared on an absorption costing basis and Saburit would like to have the statement revised...
-
When a constant force is applied to an object, the acceleration of the object varies inversely with its mass. When a certain constant force acts upon an object with mass 2 kg, the acceleration of the...
-
Use the following for all 3 circuits. V1 = 9.0 V, V = 12.0 V R = 2.0 ohms, R = 4.0 ohms, R3 = 6.0 ohms, R4 = 8.0 ohms C1 = 3.0 C = 3.0 (a) Find I in circuit A (b) Find I1 in circuit B R w R3 V R R4...
-
In Exercises 9 through 22, find the intervals of increase and decrease for the given function. h(u) = V9
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range? (a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
-
Predict the products of the following Diels?Alder reactions; (b) (a)
-
Show the structure, including stereochemistry, of the product from the following Diels?Alder reaction: COCH3 C
-
When credit terms for a sale are 2/15, n/40, the customer saves by paying early. What percent (rounded) would this savings amount to on an annual basis
-
An industrial robot that is depreciated by the MACRS method has B = $60,000 and a 5-year depreciable life. If the depreciation charge in year 3 is $8,640, the salvage value that was used in the...
-
What determines a firm's beta? Should firm management make changes to its beta? Be sure to consider the implications for the firm's investors using CAPM.

Study smarter with the SolutionInn App


