Predict the product(s) for each of the following transformations: a. b. c. d. e. f. 1) ,
Question:
a.
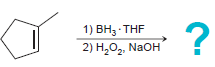
b.
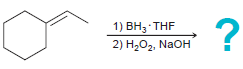
c.
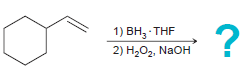
d.
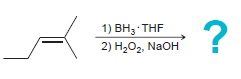
e.
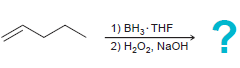
f.
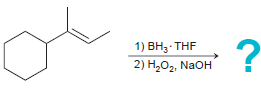
Transcribed Image Text:
1) ВН, THF 2) Н,О, NaOH 1) BH3 THF 2) H202, NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (20 reviews)
a b c d ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the product(s) for each of the following reactions. In each case, make sure to consider the number of chirality centers being formed. a. b. c. d. e. f. Os0, (catalytic) NMO 1) OsO, 2) NaHSO,...
-
Predict the major product(s) for each of the following reactions: 1) Hg(OAC)2, -0 2) NABH, ? - O, NaOH, cold Br2 H20 Pt
-
Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH 2) ,* z . Pt
-
Lacoste t-shirts come with an average price of $ 120 a piece, at their factory outlet with a std. deviation of $ 17. But at the Seasonal Sale (Discount) outlets of these t- shirts, it was also...
-
What facts may weigh in favor of or against Chou in terms of the parties' objective intent to contract?
-
In Exercises (a) Find all points of intersection of the graphs of the two equations (b) Find the unit tangent vectors to each curve at their points of intersection (c) Find the angles (0 90)...
-
3. Given a continuously compounded risk-free rate of 3% annually, at what lease rate will forward prices equal the current commodity price? (Recall the copper example in Section 3.) If the lease rate...
-
John Smith worked in the stockyard of a large building supply company. One day he unexpectedly left for California, never to return. His foreman seized the opportunity to continue to submit time...
-
A business collects cash from a customer on settlement of accounts receivable for $1500. The Journal Entry for this transaction would include a Debit to and a Credit to
-
November 1, 2015, Ralph was driving his automobile eastbound on Congress Parkway in Chicago when he became distracted by two people arguing on the sidewalk. This distraction resulted striking a large...
-
In generating electricity, how do the environmental effects of coal combustion and conventional nuclear fission compare?
-
What were the effects of the nuclear power plant accidents at Three Mile Island in Pennsylvania, Chernobyl in Ukraine, and Fukushima Daiichi in Japan?
-
The chapter contained an illustration in which the value of the information provided by a quality control system was measured using the incremental expected-payoff model of decision theory. (a) Can...
-
Skinovations needs to put together a Production schedule for next week and has asked its marketing team to give its forecasts for next week's sales. The team has used two different forecasting...
-
If a potential leader viewed her least preferred co-worker in favorable terms, how would Fiedler's Model describes this leader?
-
You have just been hired as a financial analyst for Lydex Company, a manufacturer of safety helmets. Your boss has asked you to perform a comprehensive analysis of the company s financial statements,...
-
For our first discussion you should locate a research article in which a quantitative study is reported. This article should not be a theoretical article or a methods article, but should describe...
-
A box is separated by a partition which divides its volume in the ration of 3:1. the larger portion of the box contains 1000 molecules of Ne gas; the smalled portion contains 100 molecules of He gas....
-
Granville Thomas is a citrus grower in Florida. He estimates that if 60 orange trees are planted, the average yield will be 400 oranges per tree. The average yield will decrease by 4 oranges per tree...
-
The test statistic in the NeymanPearson Lemma and the likelihood ratio test statistic K are intimately related. Consider testing H 0 : = 0 versus H a : = a , and let * denote the test statistic...
-
Predict the product of the following Diels?Alder reaction: ?
-
Draw a segment of the polymer that might be prepared from 2-phenyl-1, 3-butadiene.
-
Show the mechanism of the acid-catalyzed polymerization of 1, 3-hutadiene.
-
What is Coke's average ownership percentage in its equity method investments? Goodwill is 7000 Calculate the firm's current ratio (current assets/current liabilities). Calculate the current ratio...
-
John has to choose between Project A and Project B, which are mutually exclusive. Project A has an initial cost of $30,000 and an internal rate of return of 16 percent. Project B has an initial cost...
-
Complete the table below, for the above transactions

Study smarter with the SolutionInn App


