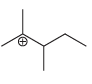
Predict whether each of the following carbocations will rearrange. If so, draw the expected rearrangement using curved
Question:
a.
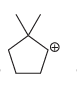
b.
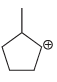
c.
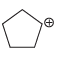
d.
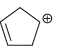
e.
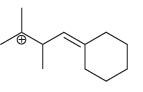
f.
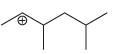
g.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a b c This carbocation is seco...View the full answer

Answered By

Michael Mulupi
I am honest,hardworking, and determined writer
4.70+
72+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether each of the following molecules is polar or non-polar: (a) IF (b) CS2 (c) SO3 (d) PCl3 (e) SF6 (f) IF5
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict whether each of the following compounds is molecular or ionic: (a) B2H6 (b) CH3OH (c) LiNO3 (d) Sc2O3 (e) CsBr (f) NOCl (g) NF3 (h) Ag2SO4.
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
Read the case "The Boys Versus Corporate" and write a short paper related to cultures
-
Find three consecutive numbers in an arithmetic sequence such that their sum is 15 and the sum of their squares is 77.
-
A machine shop has 8 identical drilling machines manned by 6 operators. The machines cannot be worked without an operator wholly engaged on it. The original cost of all these 8 machines works out to...
-
On January 1, 2010, Doone Corporation acquired 60 percent of the outstanding voting stock of Rockne Company for $300,000 consideration. At the acquisition date, the fair value of the 40 percent...
-
Please fix E10-15 Preparing a Debt Payment Schedule with the Effective-Interest Method of Amortization, and Determining Reported Amounts LO10-3 Shuttle Company issued $2,500,000, three-year, 8...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
Draw curved arrows for each step of the following mechanism: : : : 0: . : : -O: : - : : : : H20 -
-
Consider the following reaction: The following rate equation has been experimentally established for this process: Rate = k [HO - ] [CH 3 CH 2 Br] The energy diagram for this process is shown below:...
-
What is a sales mix? Provide an example of a sales mix.
-
IRIS Ratio Analysis Spreadsheet Using the information presented below, calculate the ratios requested in the IRIS ratio section. All of your answers should be in percentage-format. Note that the...
-
https://www.youtube.com/watch?v=jQbXao0mQ1M 1. Identify the cultural misunderstandings that occurred during Kenichi Takahashi's meeting with Rob, Ella, and Stephanie. Explain and support your...
-
Using what you know of groups and teams, how do you get your teams back on track with Pat? What actions do you need to take as a leader to minimize/repair the negative impact Pat's has had on the...
-
The demand function for a certain product is given by p = 3000 2x + 100 (0 x 10) where x (measured in units of a thousand) is the quantity demanded per week and p is the unit price in dollars. Sketch...
-
QUESTION 4 Dr. Martin Luther King's Speech where he proclaimed, "Free at last! Free at last!" is an example of O A. Impromptu O B. Dissolving OC. Prepared D. Reference E.Planned Speaking Wording...
-
Complete the following. (a) Graph y = f(x) in the standard viewing rectangle. (b) Approximate the coordinates of each turning point. (c) Estimate any local extrema. fx) = 4x -x =
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Compound B, an isomer of A (Problem 20.47), is also soluble in dilute HCl. The IR spectrum of B shows no bands in the 3300-3500-cm-1 region. The broadband proton-decoupled 13C spectrum of B is given...
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
Show how you might prepare each of the following amines through reductive amination: (a) (b) (c) NH2 CH3 CH3
-
3. The nominal interest rate compounded monthly when your $7,000 becomes $11,700 in eight years is ________
-
An investor can design a risky portfolio based on two stocks, A and B. Stock A has an expected return of 21% and a standard deviation of return of 39%. Stock B has an expected return of 14% and a...
-
Advanced Small Business Certifica Drag and Drop the highlighted items into the correct boxes depending on whether they increase or decrease Alex's stock basis. Note your answers- you'll need them for...

Study smarter with the SolutionInn App


