Propose a plausible synthesis for each of the following transformations: a. b. c. d. e. f. g.
Question:
a.
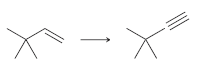
b.
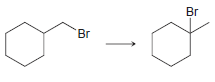
c.
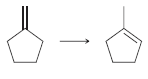
d.
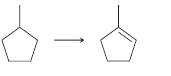
e.
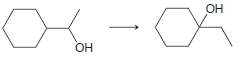
f.
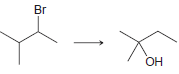
g.
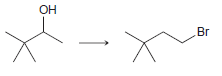
h.
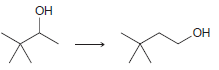
Transcribed Image Text:
Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a b c d ...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a plausible synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) Br Br
-
Propose a plausible synthesis for each of the following transformations: (a) (b) (c) (d)
-
Propose a plausible synthesis for each transformation. a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. q. r. s. t. u.
-
The regression of the X and Y equation is Y = 48.32 -2.105X, What is the slope of the data.
-
1. How would you apply the concepts of identity, power, and advocacy when working with clients of a different gender, race, ethnicity, social class, or sexual orientation? Why do you think these...
-
The spring has a stiffness k = 3 lb/ft and an unstretched length of 2 ft. If it is attached to the 5-lb smooth collar and the collar is released from rest at A, determine the speed of the collar just...
-
_____ cost are not assigned to the product but are charged as expenses against revenue. (a) Discretionary cost (b) Period cost (c) Absolute cost (d) Sunk cost
-
A fire recently destroyed a substantial portion of Manley Companys production capacity. It will be many months before capacity can be restored. During this period, demand for the firms products will...
-
if I am an accountant why would I choose the % of sales method or the % of Accounts Receivable method? What is the difference in my reasoning for choosing either or?
-
In a pretest, data on Nike were obtained from 45 respondents. These data are given in the following table, which gives the usage, sex, awareness, attitude, preference, intention, and loyalty toward...
-
Identify the reagents you would use to accomplish each of the following transformations: a. b.
-
Identify the reagents you would use to convert 2-bromo-2-methylbutane into 3-methyl-1-butyne.
-
AcelRx Pharmaceuticals, Inc., develops therapies for pain relief for a variety of patients, including cancer and trauma patients. The following data (in thousands) were adapted from recent financial...
-
Conservation efforts include reintroduction of species into the wild from captive breeding programs. Leung et al. (2018) rewilded mice from the inbred laboratory strain of mouse, C57BL/6, that had...
-
The ending balance of the Accounts Receivable account was \(\$ 7,800\). Services billed to customers for the period were \(\$ 21,500\), and collections on account from customers were \(\$ 23,600\)....
-
Cash Flow Activity Classification Classify each activity as financing, investing, or operating: 1. Repay a loan from a bank. 2. Sell merchandise from a storefront operation. 3. Dispose of an old...
-
Generally Accepted Accounting Principles Select the best answer to each of the following MBC) questions: 1. Accounting rules are developed to provide: a. Simplicity b. Useful information c....
-
Basic Accounting Principles Identify whether the following statements are true or false. 1. Together the revenue recognition principle and the expense recognition (matching) principle define the...
-
In Exercises 5358, begin by graphing f(x) = log 2 x. Then use transformations of this graph to graph the given function. What is the vertical asymptote? Use the graphs to determine each functions...
-
Complete the equations for the following equilibria and calculate Keq where the Keq expression includes [HO]. Be sure to enter Keq in proper scientific notation. (a) ammonia (acting as a base) reacts...
-
The reaction of an alkyl chloride (or bromide) with sodium iodide in acetone proceeds according to the following equation: Sodium iodide is soluble in acetone, whereas both sodium chloride and sodium...
-
The Lucas test is used to check for the presence of an alcohol functional group in an unknown compound. The test reaction is shown in the following equation: Smaller alcohols are soluble in the...
-
Explain why this secondary alcohol reacts with HCl and ZnCl2 in H2O at about the same rate as a primary alcohol (see Problem 8.38) OH Cl-CH,CHCH3
-
Show that the convexity for a zero coupon bond with m payments per year is (m) n(n + -)(1+ m m
-
Abdul Canarte , a Central Bank economist, noticed that the total group purchasing basket of goods (CPI) has gone from $149,740.00 to $344,460.00 in 8 years. With monthly compounding, what is the...
-
ABC Corporation expects sales next year to be $50,000,000. Inventory and accounts receivable (combined) will increase $8,000,000 to accommodate this sales level. The company has a profit margin of 6...

Study smarter with the SolutionInn App


