Explain why this secondary alcohol reacts with HCl and ZnCl2 in H2O at about the same rate
Question:
Explain why this secondary alcohol reacts with HCl and ZnCl2 in H2O at about the same rate as a primary alcohol (see Problem 8.38)
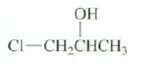
Transcribed Image Text:
OH Cl-CH,CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (12 reviews)
The carbocation which is formed at t...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A 6.11-g sample of a Cu-Zn alloy reacts with HCl acid to produce hydrogen gas. If the hydrogen gas has a volume of 1.26 L at 22C and 728 mmHg, what is the percent of Zn in the alloy? (Cu does not...
-
Explain why maleic anhydride reacts rapidly with 1,3-butadiene but does not react at all with ethene under thermal conditions. maleic anhydride
-
Explain relationships between primary and secondary boycotts.
-
The task in this design project is to design an actuation system to power the ram's reciprocating motion in a small-size shaper. The power source of the actuation system is an AC motor with 0.75 hp...
-
In each case below, fill in the missing amount: 20 000 kg Work in process, 1 April 1 Units started during April 32 000 kg Units completed during April 6000 kg Work in process, 30 April Work in...
-
On August 27, 2015, Celgene Corporation acquired all of the outstanding stock of Receptos, Inc., in exchange for $7.6 billion in cash. Referring to Celgene's 2015 financial statements and its July...
-
Compute the SD of the stock price. Interpret what this number means.
-
EnviroFriend Structures, Inc., builds environmentally sensitive structures. The companys 2018 revenues totaled $2,780 million. At December 31, 2018, and 2017, the company had, respectively, $656...
-
\ table [ [ 1 March 2 0 1 5 , \ table [ [ Machinery purchased at R 5 0 0 0 0 0 0 to manufacture ] , [ ventilators . ] , [ The total useful life of the machinery was estimated at 2 5 ] , [ years and...
-
The diagram shows a semi-circle with diameter EF of length 12 cm. Angle GEF = radians and the shaded region has an area of A cm 2 . a. Show that A = 36 + 18 sin 2. b. Given that is increasing at a...
-
The Lucas test is used to check for the presence of an alcohol functional group in an unknown compound. The test reaction is shown in the following equation: Smaller alcohols are soluble in the...
-
What reagent and solvent would you use to carry out the following transformations? -Cl -Br Ph. b) C,CH Ph CH CH OCCH3 Br Ph. Ph-C-OCH (Racemic) c) CH,CH CH-CH3 -NH,CH, C d) tom Br e) a)
-
The National Association of Realtors Existing-Home Sales Series provides a measurement of the residential real estate market. One of the measurements it produces is the Housing Affordability Index...
-
(a) Draw a simplified ray diagram showing the three principal rays for an object located inside the focal length of a converging lens, closer to the lens than to the focal point. (b) Is the image...
-
Power efficiency has become very important for modern processors, particularly for embedded systems. Create a version of gcc for two architectures that you have access to, such as x86, RISC-V,...
-
There is a movement toward wireless mobile computing using thin-client technology. Go to the Web and visit some of the ma jor computer vendors that are producing thin-client products such as handheld...
-
Draw a B-tree of order 4 and height 3 containing the fewest elements. Show an example of a split that would be applied by inserting the fewest number of elements.
-
Repeat Example 10-4, except calculate the diameter at the bottom of the column. Example 10-4 A distillation column is separating n-hexane from n-heptane using 1-in. ceramic Intalox saddles. The...
-
Compare the expenses of these functions/programs over last years figures to see the difference (last years figures should be available in last years CAFR).
-
What kind of rays are X-rays?
-
Evaluate each using the values given. (4x (y + y)); use x = -4, and y=-
-
How would you prepare the following compound using a Michaelreaction?
-
What products would result after hydrolysis from reaction of the enamine prepared from cyclopentanone and pyrrolidine with the following , -unsaturated acceptors? (a) CH 2 =CHCO 2 Et (b) H 2 C=CHCHO...
-
Show how you might use an enamine reaction to prepare each of the followingcompounds: (b) (a) CH2CH2CO2CH3 CH2CH2CN
-
Cost of debt with fees . Kenny Enterprises will issue a bond with a par value of $1,000, a maturity of twenty years, and a coupon rate of 9.9% with semiannual payments, and will use an investment...
-
Assume that an investment of $100,000 is expected to grow during the next year by 8% with SD 20%, and that the return is normally distributed. Whats the 5% VaR for the investment? A. $24,898 B....
-
Simpson Ltd is a small IT company, which has 2 million shares outstanding and a share price of $20 per share. The management of Simpson plans to increase debt and suggests it will generate $3 million...

Study smarter with the SolutionInn App


