Provide a systematic name for each of the following compounds: (a) (b) (c) (d) ci CI
Question:
(a)
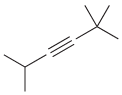
(b)
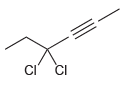
(c)
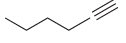
(d)
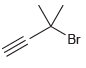
Transcribed Image Text:
ci CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
a 225trimethyl...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide a systematic name for each of the following compounds below: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q)
-
Give the systematic name for each of the following compounds a. b. (CH3)3CCH2CH2CH2CH(CH3)2 C. d. (CH3CH2)4C e. CH2CH2CH2CH2CH2NHCH2CH3 f. g. h. i. j. k. CH3OCH2CH2CH2OCH3 Br CH3HCH2CH2CHCH2CH2CH3 CH...
-
For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a. b. c. d. e. f. g. h. :N-H
-
Macmillan Learning A Geiger-Muller tube is a type gas-filled radiation detector. It can detect particles like X-rays, alpha particles, and beta rays (electrons). This is useful in quantizing the...
-
Choosing One Portfolio. The following table shows your predictions of the future returns on three mutual funds. Treasury Bills currently earn 4%. To invest all your wealth, you are limited to...
-
Use the law of cosines to solve the given problems. Find the length of the chord intercepted by a central angle of 54.2 in a circle or radius 18.0 cm.
-
What are the advantages and disadvantages of the phased approach? AppendixLO1
-
You have just been hired by EduRom Company, which was organized on January 2 of the current year. The company manufactures and sells a variety of educational DVDs for personal computers. It is your...
-
Please use Excel Formulas 7. Calculate: (a) shortage or overage (b) cumulative markup % (c) maintained markup % (d) gross margin % (12 points) Given: Cost $ 27,528 15,400 180 1,150 1,280 790 Retail $...
-
A 3-ft-diameter vertical cylindrical tank open to the atmosphere contains 0.3-m-high water. The tank is now rotated about the centerline, and the water level drops at the center while it rises at the...
-
What are some uses for hydrogen fuel cells?
-
What do operations strategies have to do with core competencies?
-
Michael and Kevin want to buy chocolates. They can't agree on whether they want chocolatecovered almonds, chocolate-covered peanuts, or chocolate-covered raisins.They agree to create a mix. They...
-
Silver Company makes a product that is very popular as a Mothers Day gift. Thus, peak sales occur in May of each year, as shown in the companys sales budget for the second quarter given below: April...
-
Among the following statements, select the ones which have a positive environmental impact. Choose several answers Minimising the impact of a product on the environment Avoiding the destruction of a...
-
Developing Financial Statements: All organizations, including those in the healthcare industry, need to make money to be profitable and survive. Financial statements, such as balance sheets, profit...
-
The engineers estimated that on average, fuel costs, assuming existing routes and number of flights stay the same, would decrease by almost 18% from an average of 42,000 gallons of jet fuel per...
-
It's the latest Berkeley trend: raising chickens in a backyard co-op coop. (The chickens cluck with delight at that joke.) It turns out that Berkeley chickens have an unusual property: their weight...
-
In Problems 1324, use the graph on the right of the function f. Is there a local maximum at 5? If yes, what is it?
-
Is times interest earned meaningful for utilities? Why or why not?
-
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system (C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of...
-
Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more strained than Cyclopropanone, is nevertheless quite stable and can even be...
-
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity of the carbonyl group into account. Cycloheptatrienone Cyclopentadienone
-
Assignment Title: The Role of Bookkeeping in Business Management and Financial Reporting Objective: Understand the importance of proper bookkeeping procedures in the management of...
-
17) The adjustment that is made to allocate the cost of a building over its expected life is called:A) depreciation expense.B) residual value.C) accumulated depreciation.D) None of the above answers...
-
9) Prepaid Rent is considered to be a(n):A) liability.B) asset.C) contra-asset.D) expense.10) As Prepaid Rent is used, it becomes a(n):A) liability.B) expense. C) contra-asset.D) contra-revenue.11)...

Study smarter with the SolutionInn App


