Ramelteon is a hypnotic agent used in the treatment of insomnia: a) What is the molecular formula
Question:
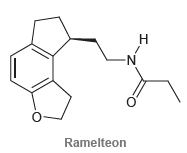
a) What is the molecular formula of this compound?
b) How many sp3-hybridized carbon atoms are present in this structure?
c) How many sp2-hybridized carbon atoms are present in this structure?
d) How many sp-hybridized carbon atoms are present in this structure?
e) How many lone pairs are present in this structure?
f) Identify each lone pair as localized or delocalized.
g) Identify the geometry of each atom (except for hydrogen atoms).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





