What is the shape of benzene, and what hydribization do you expect for eachcarbon? C=c H-C
Question:
What is the shape of benzene, and what hydribization do you expect for eachcarbon?
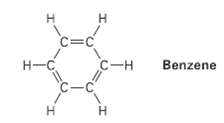
Transcribed Image Text:
C=c H-C С -н Benzene C-C I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
HC CC 11 H CH 1 C 1 H Benzen...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the shape of the yield curve given in the following term structure? What expectations are investors likely to have about future interestrates? Term 1 year 2 years 3 years 5 years 7 years 10...
-
What is the shape of the chi-square distribution?
-
What is the shape of a monopolists demand curve and marginal revenue curve?
-
Distinguish between a National Health Insurance system and a National Health Service. Provide examples of each. What kind of a system does the United States have?
-
Shondee Corporation uses the lower of cost or market and FIFO inventory methods. At the end of 2015, the FIFO cost of the ending inventory was $181,000, and the market value of the inventory was...
-
Consider the regression model fit to the nisin extraction data in Exercise 12-18. Use nisin extraction as the response. (a) Test for significance of regression using α = 0.05. What is...
-
What are the implications for the resources and capabilities the company needs?
-
Various pension plan information of the Kerem Company for 2010 and 2011 is as follows: Required Fill in the blanks lettered (a) through (k). All the necessary information is listed. It is not...
-
Please check my completed assignment for errors. Please show calculations if possible, Thank you! Record the following adjusting entries in general journal form as of December 31, 2020: 1. Supplies...
-
Patel and Sons Inc. uses a standard cost system to apply factory overhead costs to units produced. Practical capacity for the plant is defined as 55,200 machine hours per year, which represents...
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What bonds angles do you expect for each of the following, and what kind of hydribization do you expect for the central atom ineach? (a) (b) H. OH O (c) H2N-CH2-C-OH CHCH Lactic acid (in sour milk)...
-
Eliminate the parameter to find a description of the following curves in terms of x and y. Give a geometric description and the positive orientation of the curve. x = 4 cos t, y = 3 sin t; 0 t 2
-
Malaysian Agrifood Corporation Berhad reported sales of RM 7 0 , 0 0 0 in May and RM 8 0 , 0 0 0 in June. The forecast sales for July, August and September are RM 9 0 , 0 0 0 , RM 1 0 0 , 0 0 0 , and...
-
1. Refer to the \"Plotting Data\" lesson (end of "Patterns to Notice") and plot the "Skydiver Velocity vs. Time\" data (taken from the video) on the following graph. to c) d) e) f) 9) Which is the...
-
The Star Company is considering a change in its credit terms to increase sales. Its current credit sales are $8.5 million per year and its present credit terms are 2/16 net of 20 basis. Discount is...
-
Determine the shear flow distribution for a torque of 57265.0Nm applied to the three cell section shown in the figure below. Note that the section has a constant shear modulus throughout. All answers...
-
2 a) Figure 1 shows the orientation of the carbon fibre which is orientate at 0.A9 radians from the vertical plane. Draw and label the element's normal and shear stresses acting on the carbon fibre...
-
Which statement is most appropriate? a. Variety creates a richness and degree of flexibility in the type of audit work that is undertaken. In many cases, an audit department will contain different...
-
Classify each of the following as direct costs or indirect costs of operating the Pediatrics ward for children at the Cleveland Clinic: a. Wi-Fi covering the entire hospital campus b. Net cost of...
-
Proteins can exist in one of two states, the active, folded state and the inactive, unfolded state. Protein folding is sometimes thought of as a first-order phase transition from folded to unfolded...
-
Draw the structures of the compounds represented by these models and explain which would have the larger solubility in water. (The red atoms are oxygens.)
-
Draw the structures of the compounds represented by these models and explain which would have the higher melting point.
-
These models show the three isomers of C5H12. Explain which isomer would produce only one C5H11Cl isomer on reaction with Cl2 in the presence of light.
-
Jennifer purchased a home for $1,000,000 in 2016. She paid $200,000 cash and borrowed the remaining $800,000. This is Jennifer's only residence. Assume that in year 2024, when the home had...
-
business plan describing company with strengths and weaknesses. Any gaps in plan. Recommendations for improvement of the plan.
-
You wish to buy a car today for $35,000. You plan to put 10% down and finance the rest at 5.20% p.a. for six years. You will make equal monthly payments of $_______.

Study smarter with the SolutionInn App


