Rank the signals of the following compound in terms of increasing chemical shift. Identify the proton(s) giving
Question:
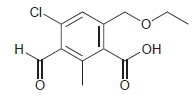
Transcribed Image Text:
CI. Н. но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (22 reviews)
10 ppm 658 ppm ...View the full answer

Answered By

Muhammad Mahtab
everyone looks that their work be perfect. I have more than a five year experience as a lecture in reputable institution, national and international. I provide perfect solution in marketing, case study, finance problems, blog writing, article writing, business plans, strategic management, human resource, operation management, power point presentation and lot of clients need. Here is right mentor who help clients in their multi-disciplinary needs.
5.00+
3+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound contains three nitrogen atoms. Rank them in order of increasing basicity.
-
The following compound has four aromatic rings. Rank them in terms of increasing reactivity toward electrophilic aromatic substitution.
-
The following compounds can all react as acids. (a) For each compound, show its conjugate base. Show any resonance forms if applicable. (b) Rank the conjugate bases in the order you would predict,...
-
Date 1 July 2019 1 June 2020 30 June 2020 1 July 2020 1 July 2020 30 June 2021 1 July 2021 Particulars (???) (???) (To record acquisition of delivery truck) (???) (???) (???) (To record minor repair...
-
What are three applications of IoTs?
-
The Excel file Retail Survey provides data about customers preferences for denim jeans. Use PivotTables and slicers to draw conclusions about how preferences differ by gender and age group and...
-
What are the four main types of consumer products?
-
Myles Company expects to produce 1,200,000 units of Product XX in 2012. Monthly production is expected to range from 80,000 to 120,000 units. Budgeted variable manufacturing costs per unit are:...
-
Using forms 1120 and 1125A in completion: 1. Prepare the 2018 Corporate Tax Return for Barbie's Closet 2. Barbara " Barbie" Robert's started a personal shopping company, Barbie's Closet. It operates...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
How many signals do you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) Geraniol Isolated from roses and used in perfumes . NH2 Dopamine A neurotransmitter that is...
-
Predict the expected number of signals in the 13 C NMR spectrum of each of the following compounds. For each signal, identify where you expect it to appear in the 13 C NMR spectrum: (a) (b) (c)
-
NTFS file records are made up of components which are also called attributes, although the word has a different meaning. What is an NTFS attribute? What are the contents of an NTFS data attribute?
-
Repeat Prob. 10-18 for signed-magnitude binary numbers. Prob. 10-18 Derive an algorithm in flowchart form for the comparison of two signed binary numbers when negative numbers are in signed-2's...
-
Tideview Home Health Care, Inc., has a bond issue outstanding with eight years remaining to maturity, a coupon rate of 10 percent with interest paid annually, and a par value of $1,000. The current...
-
Captain Billy Whirlywhirl Hamburgers issued 7%, 10-year bonds payable at 70 on December 31, 2010. At December 31, 2012, Captain Billy reported the bonds payable as follows: Captain Billy Whirlywhirl...
-
Two scenarios about the future of the global economy in 2050 have emerged. Known as continued globalization, the first scenario is a (relatively) rosy one. Spearheaded by Goldman Sachs, whose...
-
Visit www.pearsonglobaleditions.com/malhotra to read the video case and view the accompanying video. Nike: Associating Athletes, Performance, and the Brand highlights Nike's use of marketing research...
-
Select an appropriate type of modeling function for the data shown in the graph. Choose from the following. i. Exponential ii. Logarithmic iii. Logistic
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Using 13 CO 2 as your only source of labeled carbon, along with any other compounds needed, how would you synthesize the following compounds? (a) CH 3 CH 2 13 CO 2 H (b) CH 3 13 CH 2 CO 2 H
-
How would you carry out the following transformations? "Co CH2 C Lc
-
Which method?Grignard carboxylation or nitrile hydrolysis?would you use for each of the following reactions? Explain. " - C CH2Br (a) " CH Br (b) CH3CH2CHCO2H CCH2CHCH3 (c) CHH-H2H2, CHH-H-CH21...
-
This short exercise demonstrates the similarity and the difference between two ways to acquire plant assets. (Click the icon to view the cases.) Compare the balances in all the accounts after making...
-
Balance sheet and income statement data for two affiliated companies for the current year appear below: BALANCE SHEET As at December 31, Year 6 Albeniz Bach Cash $ 40,000 $ 21,000 Receivables 92,000...
-
please reference excel cells Caroll Manufacturing company manufactures a single product. During the past three weeks, Caroll's cost accountant observed that output costs varied considerably. The...

Study smarter with the SolutionInn App


