Alka-Seltzer is an over-the-counter medicine used to treat acid indigestion and heartburn. The active pharmaceutical ingredients (API)
Question:
Alka-Seltzer® is an over-the-counter medicine used to treat acid indigestion and heartburn. The active pharmaceutical ingredients (API) in each Alka-Seltzer® tablet are aspirin (325 mg), citric acid (1.000x103 mg), and sodium bicarbonate (1.916x103 mg). For a single dose, two Alka-Seltzer® tablets are dissolved in 4.0 fluid ounces of water (1.0 fluid ounce = 29.57 mL), causing the following reaction:
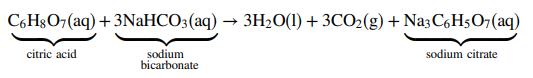
(a) What volume of CO2 gas (mL) at 25°C and 1 atm would be produced by a normal dose of Alka-Seltzer®? Assume ideal-gas behavior and that the reaction goes to completion.
(b) You wake up feeling miserable before your material-and-energy-balances final exam, but you know you can’t miss it. You grab some Alka-Seltzer® and an 11-ounce bottle of water. You drink just enough water to leave 4.0 fluid ounces in the bottle (you’ve had lots of practice with this). Then you drop in the two tablets, screw the cap tightly onto the bottle, and rush out the door. Calculate the pressure inside the bottle, assuming the temperature remains constant at 25°C and neglecting the volume of the tablets.
(c) How reasonable are the assumptions of ideal-gas behavior in Parts (a) and (b)?
Step by Step Answer:

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard





