The data in the following table have been obtained for the potential of the cell Pt(s) |H
Question:
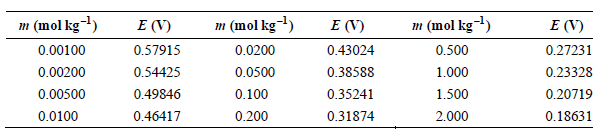
The data in the following table have been obtained for the potential of the cell Pt(s) |H2(g, f = 1atm) HCl(aq, m)|AgCl(s) Ag(s) as a function of m at 25°C.
a. Determine E° using a graphical method.
b. Calculate γ ± for HCl at m = 0.00100, 0.0100, and 0.100 mol kg–1.
Transcribed Image Text:
m (mol kg) m (mol kg) m (mol kg-l) E (V) 0.57915 0.54425 0.49846 E (V) E (V) 0.27231 0.23328 0.20719 0.500 0.00100 0.0200 0.43024 0.38588 0.35241 0.00200 0.0500 1.000 0.00500 0.0100 1.500 2.000 0.100 0.200 0.46417 0.31874 0.18631
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
Cell reaction 2AgCls H 2 g2Ags 2H aq 2Cl aq In the low ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following data have been obtained for oxygen gas at 273.15 K. Calculate the best value of the gas constant R from them and the best value of the molar mass of02 0.750000 0.250000 Platm 0.500000...
-
The following data have been obtained for the liquid-vapour equilibrium compositions of mixtures of nitrogen and oxygen at 100 kPa. T/K 77.3 78 80 82 84 86 88 90.2 X (O2) 0 10 34 54 70 82 92 100...
-
Your task in this problem will be to use a spreadsheet to generate a Txy diagram for a two-component system, using Raoult?s law to express the vapor?liquid equilibrium distribution of each species....
-
A problem in Statistics is given to three students A, B, and C whose chances of solving it are 1/4 and 1/5 respectively. Find the probability that the problem will be solved if they all try...
-
Discuss the key ethical issues of taste, stereotyping, use of sex appeal, and targeting children. How do advertising professionals deal with these issues?
-
What is a quadratic equation?
-
Imagine that you designed a high-end line of clothing and accessories. Which factors should you consider when choosing retail partners? What type of retailer would you choose as a partner? Why?
-
The controller of Arrowroot Company wishes to improve the companys control system by preparing a monthly cash budget. The following information relates to the month ending July 31, 20X1: June 30,...
-
Urwick Company purchased inventory for $30,000 on account; terms are 3/10, n/30. If $6,000 of the inventory was returned and the remaining amount due was paid within the discount period, the purchase...
-
The magnetic circuit of figure consists of a core and a moveable plunger of width l p , each of permeability ?. The core has cross-sectional area A and means length A c . The overlap area of the two...
-
Show that the estimates Î²Ì 1 = 1 . 572 and Î²Ì 2 = 1 . 357 used in the first experiment of Table 3.1 are in fact the OLS estimators. Table 3.1 Y2i (6) Y; X (2) i...
-
What is the mean of X? One interesting phenomenon of bacteriuria is that there is a turnover; that is, if bacteriuria is measured on the same woman at two different points in time, the results are...
-
What factors help make workers in high-income economies more productive than workers in other economies?
-
Problem 1 PROBLEMS Sabres Limited, a Canadian-controlled private corporation whose fiscal year end is December 31, provides you with the following data concerning its tax accounts and capital...
-
9.6. A habitual gambler often visits three different casinos and plays roulette there. He wants to discover at which casino he has better luck with his roulette games. So, he records his gambling...
-
The firm has estimated that its sales for 2 0 1 3 will be $ 8 4 6 , 7 5 6 Cash dividends to be paid by the firm in 2 0 1 3 $ 3 7 , 7 2 0 Minimum cash balance to be maintained by the firm $ 2 8 , 5 1...
-
Bob Long was hired by County Hospital aS supervisor of engineering and maintenance. Although well experienced in his field, this was his first management job. Soon after Bob's arrival a maintenance...
-
Initial Outlay (IO) 1. A company is considering purchasing a machine for $100,000. Shipping costs would be another $5,000. The project would require an initial investment in net working capital of...
-
In Problems 4374, find the real solutions of each equation. 1 (x - 1) 1 x - 1 12
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Discuss the physical origins of the surface Gibbs energy.
-
The rate of sedimentation of a recently isolated protein was monitored at 20C and with a rotor speed of 50 000 r.p.m. The boundary receded as follows: Calculate the sedimentation constant and the...
-
Find the drift speed of a particle of radius 20 m and density 1750 kg m 3 which is settling from suspension in water (density 1000 kg m 3 ) under the influence of gravity alone. The viscosity of...
-
Comfort Golf Products is considering whether to upgrade its equipment Managers are considering two options. Equipment manufactured by Stenback Inc. costs $1,000,000 and will last five years and have...
-
Weaver Corporation had the following stock issued and outstanding at January 1, Year 1: 71,000 shares of $10 par common stock. 8,500 shares of $60 par, 6 percent, noncumulative preferred stock. On...
-
Read the following case and then answer questions On 1 January 2016 a company purchased a machine at a cost of $3,000. Its useful life is estimated to be 10 years and then it has a residual value of...

Study smarter with the SolutionInn App


