The unimolecular decomposition of urea in aqueous solution is measured at two different temperatures and the following
Question:
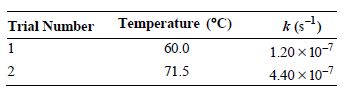
a. Determine the Arrhenius parameters for this reaction.
b. Using these parameters, determine ΔH€¡ and ΔS€¡ as described by the Eyring equation.
Transcribed Image Text:
Temperature (°C) k (s-) Trial Number 60.0 1.20 x 10-7 71.5 4.40 x 10-7 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a The activation energy is determined by taking the ratio of the rate constants ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Hydrogen abstraction from hydrocarbons by atomic chlorine is a mechanism for Cl · loss in the stratosphere. Consider the reaction of Cl· with ethane: C 2 H 6 (g) + Cl· (g) C 2 H...
-
Suppose the vapor pressure of a substance is measured at two different temperatures. (a) By using the Clausius-Clapeyron equation (Equation 11.1) derive the following relationship between the vapor...
-
The gas-phase decomposition of ethyl bromide is a first-order reaction, occurring with a rate constant that demonstrates the following dependence on temperature: a. Determine the Arrhenius parameters...
-
why is teamwork so important especially in healthcare? explain
-
Find (a) the domain and (b) the zeroes of the function. 1. f (x) = (x -5) / (2x2 - x) 2. f (x) = 10 - (3 - x)
-
In Problems 91 106, find the quotient and the remainder. Check your work by verifying that (Quotient )(Divisor ) + Remainder = Dividend 3x 3 x 2 + x 2 divided by x + 2
-
Describir las principales funciones de la administracin de ventas .
-
Jessica Company manufactures hockey pucks and soccer balls. For both products, materials are added at the beginning of the production process and conversion costs are incurred evenly. Jessica uses...
-
An amount is deposited for eight years at 20%. If compounding occurs quarterly, then the table value is found at 5% for eight periods. 20% for 32 periods. 20% for eight periods. 5% for 32 periods. 1
-
For the payoff table below, the decision maker will use P(s1)=.15; P(s2)=.5; P(s3)=.35 STATE OF NATURE a. What alternative would be chosen according to expected value? b. For a lottery having a...
-
Catalase is an enzyme that promotes the conversion of hydrogen peroxide (H 2 O 2 ) into water and oxygen. The diffusion constant and radius for catalase are 6.0 10 7 cm 2 s 1 and 51.2 ,...
-
Consider the unimolecular isomerization of methylcyanide, a reaction that will be discussed in detail in Chapter 36: CH 3 NC(g) CH 3 CN(g) The Arrhenius parameters for this reaction are A = 2.5 10...
-
Sapient Corporation is a technology consultancy firm discussed in Problem 6.15 (Chapter 6) in relation to unusual charges reported for Year 2 and Year 3. For Year 3, the firm's Form 10-K filing...
-
Create your own privacy philosophy. This should cover the policies you will use for email, texting, social media, and internet usage. Consider what information is being collected anout you in each...
-
1. What future markets might be attractive to Carrefour and which mode of operation would be preferable? How important is the theoretical concept of psychological distance? 2. Corporate...
-
Briefly restate your problem space and methodology. Considering your problem space and methodology, what factors are you considering in deciding whether to use a theoretical foundation or a...
-
In light of your personal experience, what strategies or approaches do you believe could be effective in creating a workplace environment where employees from diverse cultural backgrounds feel both...
-
1. Entrepreneurs hold many common traits, identify five common traits of an entrepreneur that resonate with you and discuss each one of the five traits and why they matter to you. 2. Why is...
-
Two functions, f and g, are related by the given equation. Use the numerical representation of f to make a numerical representation of g. g(x) = f(x + 1) - 2 X f(x) 1 2 2 4 3 3 4 7 5 8 00 6 10
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
At 298 K and 1 bar pressure, the density of water is 0.9970 g cm 3 , and C P,m = 75.3 J K -1 mol -1 . The change in volume with temperature is given by V = V i T, where , the coefficient of thermal...
-
5-Methylcyclopentadiene undergoes homolytic bond cleavage of a C!H bond to form a radical that exhibits five resonance structures. Determine which hydrogen is abstracted and draw all five resonance...
-
Calculate S surroundings and S total for the processes described in parts (a) and (b) of Problem P5.16. Which of the processes is a spontaneous process? The state of the surroundings for each part is...
-
Slow Roll Drum Co. is evaluating the extension of credit to a new group of customers. Although these customers will provide $198,000 in additional credit sales, 13 percent are likely to be...
-
Wendell's Donut Shoppe is investigating the purchase of a new $39,600 conut-making machine. The new machine would permit the company to reduce the amount of part-time help needed, at a cost savings...
-
1.Discuss the challenges faced with Valuing Stocks and Bonds. As part of this discussion, how will the selected item be implemented in an organization and its significance? 2. Discuss how Valuing...

Study smarter with the SolutionInn App


