The gas-phase decomposition of ethyl bromide is a first-order reaction, occurring with a rate constant that demonstrates
Question:
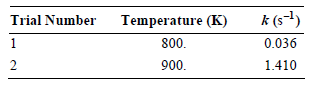
a. Determine the Arrhenius parameters for this reaction.
b. Using these parameters, determine ΔH€¡ and ΔS€¡ as described by the Eyring equation.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





