0.10 mol of nitrogen gas follow the two processes shown in Figure P19.58. How much heat is...
Question:
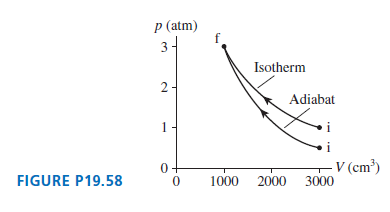
Transcribed Image Text:
p (atm) 3 Isotherm 2- Adiabat V (cm³) 3000 FIGURE P19.58 1000 2000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Model We have an adiabatic and an isothermal process Solve For the adiabatic process no heat is add...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
How much heat is required to change a 1 kg block of ice at 10oC to steam at 110oC? Give your answer in Joule and calorie and Calorie. (1 cal = 4.186 J; 1 Calorie = 1000 calorie.
-
How much heat is required to melt a 2.5-kg block of ice at 0oC?
-
How much heat is required to boil away 1.50 kg of water that is initially at 100oC?
-
Attlee Ltd holds 28% of the issued shares of Nehru Ltd. Attlee Ltd acquired these shares on 1 July 2019 and on this date all the identifiable assets and liabilities of Nehru Ltd were recorded at...
-
Blue Company, an architectural firm, has a bookkeeper who maintains a cash receipts and disbursements journal. At the end of the year (2016), the company hires you to convert the cash receipts and...
-
What is critical chain scheduling, and how is it different from critical path analysis? Give an example of its use.
-
What are the advantages and disadvantages of certified environmental product declarations? Assuming that you are the environmental manager of Global Autos, with companies operating in Sweden,...
-
At the beginning of 2014, Florida Rock Industries had 25,000 shares of common stock issued and outstanding and 500 $1,000, 6% bonds, each convertible into 10 shares of common stock . During 2014,...
-
Question 2: (Everything to be done by hand expect if otherwise is stated) The following data are provided: X Y a/a 1 2 3 4 5 6 7 8 9 10 11 12 13 35.3 29.7 30.8 58.8 61.4 71.3 74.4 76.7 70.7 57.5 46.4...
-
Chariot. com needs $ 500,000 in venture capital to bring a new Internet messaging service to market. The firms management has approached Route 128 Ventures, a venture capital firm located in the...
-
0.10 mol of nitrogen gas follow the two processes shown in Figure EX19.31. How much heat is required for each? p (atm) 3 2 - V (cm) 0+ 2000 3000 1000 FIGURE EX19.31
-
You come into lab one day and find a well-insulated 2000 mL thermos bottle containing 500 mL of boiling liquid nitrogen. The remainder of the thermos has nitrogen gas at a pressure of 1.0 atm. The...
-
A national homebuilder builds singlefamily homes and condominiumstyle townhouses. The Excel file House Sales provides information on the selling price, lot cost, type of home, and region of the...
-
There are emails, newsletters, and short progress notes that teachers can use to communicate with parents daily or weekly. What are some other ways to physically involve parents in classroom...
-
Yancey Productions is a film studio that uses a job-order costing system. The company's direct materials consist of items such as costumes and props. Its direct labor includes each film's actors,...
-
Stakeholders are the individuals and groups that are affected by and can affect what a team does or produces. A team's stakeholders usually present the team with a variety of wonderful opportunities...
-
Compare and contrast the organizational structure of theHarley Davisons and Toyotafirms and discuss the advantages and disadvantages of each structure. Web pages:...
-
Describe the results of implementing the lesson/activity with the students, including student assessment results. Discuss the long-term results you would expect to see if you differentiated the...
-
Are the present-day continents a permanent feature on our planet? Discuss why or why not.
-
Baxter, Inc., owns 90 percent of Wisconsin, Inc., and 20 percent of Cleveland Company. Wisconsin, in turn, holds 60 percent of Clevelands outstanding stock. No excess amortization resulted from these...
-
What do radio waves and light have in common? What is different about them?
-
When you look downward at a fish in water, does refraction make the fish appear closer to the surface or deeper?
-
When you view the Sun near the horizon, does refraction make it appear higher or lower in the sky?
-
provide a brief overview of the situation and, using the key concepts from BlewMinds leveraging influencer case study, the steps that can be taken to remedy the issues. The goal of the presentation...
-
Question 5 Brintex Distributors Income Statement for the year ended 30 June 20X1 Revenue: Sales Revenue $900,000 Dividends Received 3,000 Interest Earned 6,000 Profit on Sale of Plant 31,000 Total...
-
Suppose that the installed cost of a commercial solar hot water heating system is $ 1 5 , 0 0 0 and the annual fuel cost saving is $ 1 , 0 0 0 . Assume ( counter factually ) that fuel cost savings...

Study smarter with the SolutionInn App


