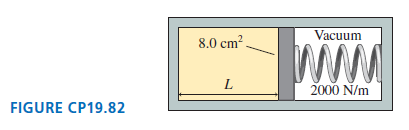
A monatomic gas fills the left end of the cylinder in Figure CP19.82. At 300 K, the
Question:
Transcribed Image Text:
8.0 cm?. Vacuum 2000 N/m FIGURE CP19.82
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Model Assume the gas is ideal Solve Apply the first law of thermodynamics Eth W Q nC V T Consider ad...View the full answer

Answered By

Joseph Njoroge
I am a professional tutor with more than six years of experience. I have helped thousands of students to achieve their academic goals. My primary objectives as a tutor is to ensure that students do not have problems while tackling their academic problems.
4.90+
10+ Reviews
27+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Helium (He), a monatomic gas, fills a 0.010-m3 container. The pressure of the gas is 6.2 105 Pa. How long would a 0.25-hp engine have to run (1 hp = 746 W) to produce an amount of energy equal to...
-
A monatomic gas and a diatomic gas both have n moles and are at temperature T. What is the difference in their internal energies? Express your answer in n, R, and T.
-
Repeat Problem 72 for a monatomic gas. B. D.
-
Generics, Inc. is a U. S. GAAP reporter that manufactures and sells generic drugs and has a December 31 year-end. On March 1, 2014, it began selling a drug, Anocyn, which is a generic of Dicital....
-
On August 10, 2014, Jasper purchased business equipment for $40,000. On his 2014 tax return, $40,000 of 179 immediate expense was taken on the equipment. On July 14, 2015, Jasper sold the equipment...
-
Kyle believes the price of Ajax stock is about to decrease. If he wants to profit from the decline in price, he should on Ajax stock. A . sell a put B . buy a call C . buy a put D . write a put
-
Explain the accounting for unexpected gains and losses.
-
A 3.00-m-long, 240-N, uniform rod at the zoo is held in a horizontal position by two ropes at its ends (Fig. 11.30). The left rope makes an angle of 150° with the rod and the right rope makes an...
-
The La Pann Company has obtained the following sales forecast data: Cash Sales Credit Sales July $80,000 240,000 August $70,000 220,000 September $50,000 180.000 October $60,000 200.000 The regular...
-
You are the owner of a small business that manages the program, food, and merchandise sales at the Excel Center, where the local professional basketball team plays. In addition to basketball games,...
-
0.020 mol of a diatomic gas, with initial temperature 20C, are compressed from 1500 cm 3 to 500 cm 3 in a process in which pV 2 = constant. How much heat energy is added during this process?
-
Solids and liquids resist being compressed. They are not totally incompressible, but it takes large forces to compress them even slightly. If it is true that matter consists of atoms, what can you...
-
Earl Tetley worked as a payroll clerk for Tinwings Inc. Over the course of several months, he defrauded the company of about $250 000. His plan was simple. He drafted cheques, on behalf of the...
-
STAR Co. provides paper to smaller companies whose volumes are not large enough to warrant dealing directly with the paper mill. STAR receives 100-feet-wide paper rolls from the mill and cuts the...
-
2. Define management.3) Describe who managers are and where they work.4) Describe what managers do.5) Describe the factors that are reshaping and redefiningmanagement.6) Explain what the external...
-
E 1 0 . 1 9 ( LO 3 ) ( Depletion Computations - Timber ) Stanislaw Timber Company owns 9 , 0 0 0 acres of timberland purchased in 2 0 1 4 at a cost of $ 1 , 4 0 0 per acre. At the time of purchase,...
-
Question 01 You were hired as an engineer to evaluate some soil characteristics and conditions to which it is subjected. To do this, you obtained the information presented in the stratified soil...
-
1 According to Strudler, the reason lying is worse than misleading is thatl ying damages trust whereas misleading does not damage any trust. According to Strudler, the reason lying is worse than...
-
The Hubble space telescope is within a cylinder 4.3 m in diameter and 13 m long. What is the volume within this cylinder?
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
What might the spectrum of an atom look like if the atoms electrons were not restricted to particular energy levels?
-
Some older cars vibrate loudly when driving at particular speeds. For example, at 65 mph the car may be most quiet, but at 60 mph the car may rattle uncomfortably. How is this analogous to the...
-
Place the proper number of electrons in each shell: Rubidium, Rb Sodium, Na Krypton, Kr Chlorine, CI
-
A contractor constructed a house for resale, which was sold immediately. For tax purposes, this is an example of A) capital income. B) business income. C) other income. D) property income.
-
You invest $100 in a risky asset with an expected rate of return of 0.12 and a standard deviation of 0.15 and a T-bill with a rate of return of 0.05. What percentages of your money must be invested...
-
Nanometrics, Inc., has a beta of 3.43. If the market return is expected to be 13.50 percent and the risk-free rate is 7.00 percent, what is Nanometrics required return? (Round your answer to 2...

Study smarter with the SolutionInn App


