The heat engine shown in FIGURE P21.63 uses 0.020 mol of a diatomic gas as the working
Question:
The heat engine shown in FIGURE P21.63 uses 0.020 mol of a diatomic gas as the working substance.
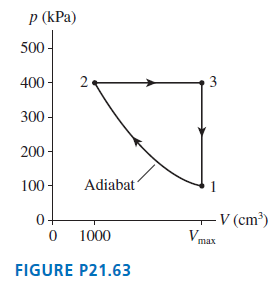 a. Determine T1, T2, and T3.b. Make a table that shows ΔEth, Ws, and Q for each of the three processes.c. What is the engine??s thermal efficiency?
a. Determine T1, T2, and T3.b. Make a table that shows ΔEth, Ws, and Q for each of the three processes.c. What is the engine??s thermal efficiency?
Transcribed Image Text:
Р (КРа) 500 - 400 - 2. 3 300 - 200 - 100- Adiabat – (cm³) Vmax 1000 FIGURE P21.63
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Model The closed cycle in this heat engine includes adiabatic process 1 2 ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
The heat engine shown in FIGURE P21.62 uses 2.0 mol of a monatomic gas as the working substance. a. Determine T 1 , T 2 , and T 3 . b. Make a table that shows ÎE th , W s , and Q for each of...
-
A heat engine using a monatomic gas follows the cycle shown in FIGURE P21.58. a. Find W s , Q, and E th for each process in the cycle. Display your results in a table.b. What is the thermal...
-
(a) Calculate the total rotational kinetic energy of the molecules in 1.00 mol of a diatomic gas at 300 K. (b) Calculate the moment of inertia of an oxygen molecule (O2) for rotation about either the...
-
Adrian Webb bought 618 bricks to build a barbecue pit, paying $185.40. Find the cost per brick. (HINT: Cost per brick means the cost for one brick.) The cost per brick is s (Round to nearest hundidth)
-
Who in a firm should be responsible for industry analysis? Assume that the firm does not have a strategic planning department.
-
Organic, vegan and eco-friendly have all become the most common terms in the personal care and cosmetic industry, but halal is the latest and emerging trend. Not so long ago, the word halal meant an...
-
Using the PewSocialMedia dataset, get a 95% confidence interval on the variable Index of Social Media Use for three groups: those who live in urban areas, those who live in suburban areas, and those...
-
On December 31, 2012, Firth Company borrowed $62,092 from Paris Bank, signing a 5-year, $100,000 zero-internet-bearing note. The note was issued to yield 10% interest. Unfortunately, during 2012,...
-
Determinants of Interest Rate for Individual Securities The Wall Street Journal reports that the rate on 3-year Treasury securities is 6.55 percent, and the 6-year Treasury rate is 6.75 percent. From...
-
At the beginning of the fall semester, Beaty State University, a public university, billed tuition and fees at a gross amount of $2,400,000. Of this amount, the University awarded scholarships of...
-
A heat engine using 120 mg of helium as the working substance follows the cycle shown in FIGURE P21.61. a. Determine the pressure, temperature, and volume of the gas at points 1, 2, and 3.b. What is...
-
A heat engine with 0.20 mol of a monatomic ideal gas initially fills a 2000 cm 3 cylinder at 600 K. The gas goes through the following closed cycle: Isothermal expansion to 4000 cm 3 . Isochoric...
-
Suppose the rate of growth of the population of a city is predicted to be dp/dt = 2000t 1.04 where p is the population and t is the number of years past 2018. If the population in the year 2018 is...
-
Investigate the Mercedes Benz company and you have to cover this topic " For Business prospects, Market growth, Market quality, and Environmental aspects are three most important factors. Explain...
-
The case study for Goodwill Industries and how they "do good" as a core business strategy. What are Goodwill's competitive advantages? Goodwill has found success in social services. What problems...
-
Cosmic Cals (Pty) Ltd , a seller of personalized scientific calculators, had an inventory of 40 calculators. The value of these calculators is R15 400 each on the 1 January 2022. During the current...
-
Perform an analysis of Best Buy Co. Inc. Your analysis will draw on the Form 10K (as of February 2013). Your analysis can include information prior to February 2013 but should not draw on any...
-
Research organizational structure of a company of your choice. Use your understanding of organizational structure to analyze whether this organization's structure is the best choice for the business...
-
In Exercises use geometric series or the nth-Term Test to determine the convergence or divergence of the series. (0.36) n=0
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
What is the ratio of the Earths orbital period about the sun to the Earths period of rotation about its own axis?
-
Joe has a weight of 720 N (about 162 lb) when he is standing on the surface of the Earth. What would his weight (the gravitational force due to the Earth) be if he doubled his distance from the...
-
Two masses are attracted by a gravitational force of 0.36 N. What will the force of attraction be if the distance between the two masses is tripled?
-
Use the following information: \ table [ [ Country , \ table [ [ Consumer Prices ] ] , Interest Rates,Current Units ( per US$ ) ] , [ Forecast , 3 - month, 1 - yx Covt Bond,, ] , [ 2 0 2 4 e ,...
-
Year-to-date, Yum Brands had earned a 3.70 percent return. During the same time period, Raytheon earned 4.58 percent and Coca-Cola earned 0.53 percent. If you have a portfolio made up of 40 percent...
-
Rate of Return If State Occurs State of Probability of Economy State of Economy Stock A Stock B Stock C Boom .15 .31 .41 .21 Good .60 .16 .12 .10 Poor .20 .03 .06 .04 Bust .05 .11 .16 .08 a. Your...

Study smarter with the SolutionInn App


