Question: A heat engine using a monatomic gas follows the cycle shown in FIGURE P21.58. a. Find W s , Q, and E th for each
A heat engine using a monatomic gas follows the cycle shown in FIGURE P21.58.
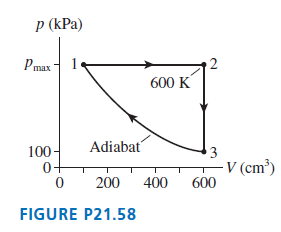
a. Find Ws, Q, and ΔEth for each process in the cycle. Display your results in a table.b. What is the thermal efficiency of this heat engine?
p (kPa) Pmax 2 600 K 100- Adiabat 3 - V (cm) 600 0+ 200 400 FIGURE P21.58
Step by Step Solution
3.45 Rating (171 Votes )
There are 3 Steps involved in it
Model For the closed cycle of the heat engine process 1 2 is isobaric process 2 3 is isochoric and p... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
1442_6054778b9d38e_693678.pdf
180 KBs PDF File
1442_6054778b9d38e_693678.docx
120 KBs Word File


