Answered step by step
Verified Expert Solution
Question
1 Approved Answer
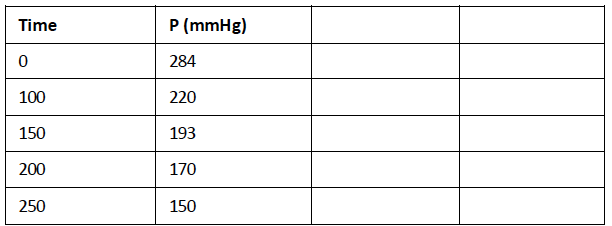
10) The rate of decomposition of azomethane (C 2 H 6 N 2 ) was studied by monitoring the partial pressure of the reactant as
10) The rate of decomposition of azomethane (C2H6N2) was studied by monitoring the partial pressure of the reactant as a function of time. Determine if the data below support a first-order reaction. Calculate the rate constant for the reaction
Thinking, the integrated method for determining the order of reaction, find ln P, 1/P, check the link to t and find the slope of -k
Time P(mmHg) 0 284 100 220 150 193 200 170 250 150Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


