Answered step by step
Verified Expert Solution
Question
1 Approved Answer
3. In the following table, indicate the orbital corresponding to the given combination of quantum numbers. If the combination of quantum numbers is not allowed,
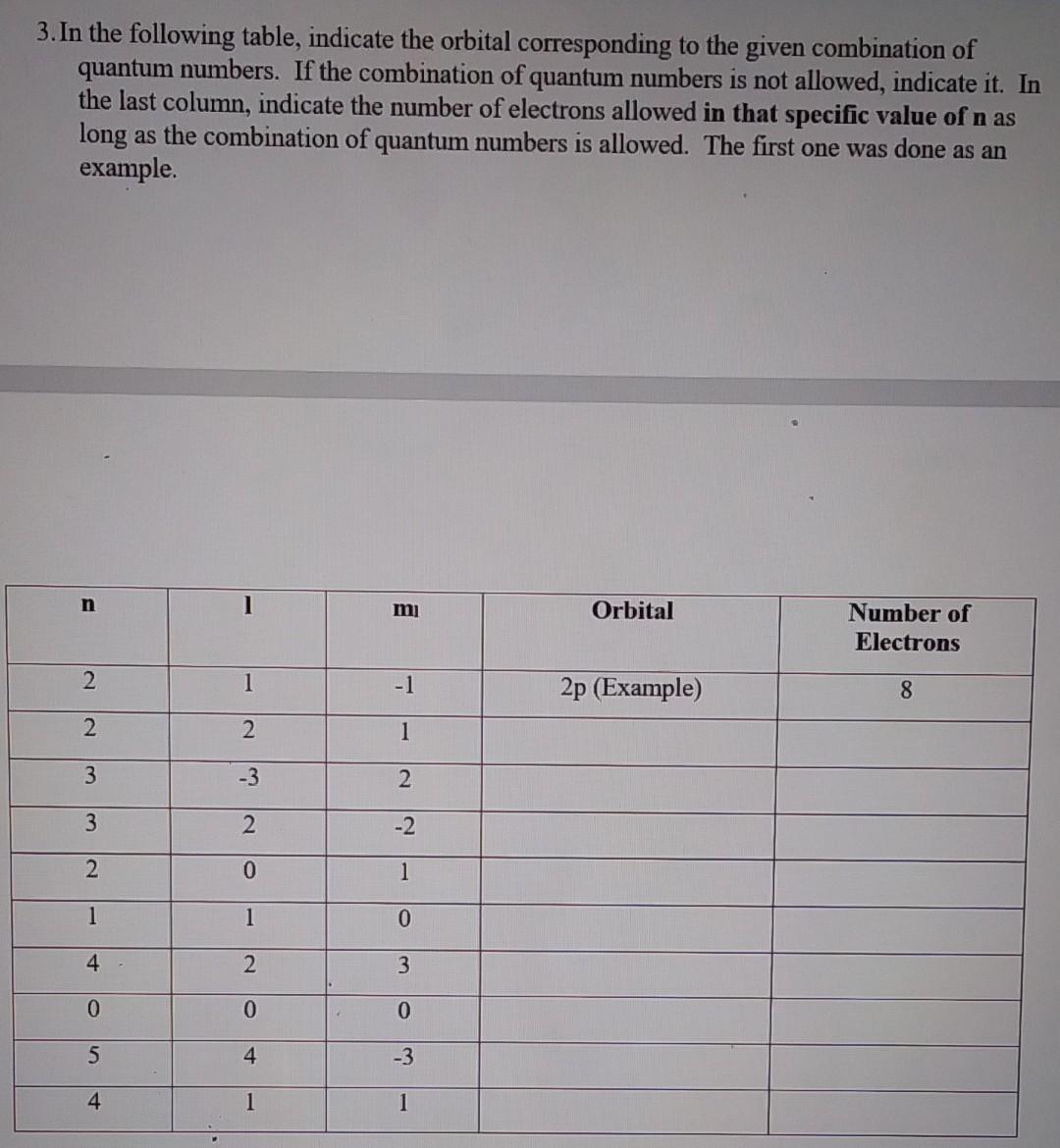
3. In the following table, indicate the orbital corresponding to the given combination of quantum numbers. If the combination of quantum numbers is not allowed, indicate it. In the last column, indicate the number of electrons allowed in that specific value of n as long as the combination of quantum numbers is allowed. The first one was done as an example. n 1 mi Orbital Number of Electrons 2 1 -1 2p (Example) 8 2 2 1 3 -3 2 3 2. -2 2 0 1 1 1 0 4 2 3 0 0 0 5 4 -3 4 1 1
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


