Answered step by step
Verified Expert Solution
Question
1 Approved Answer
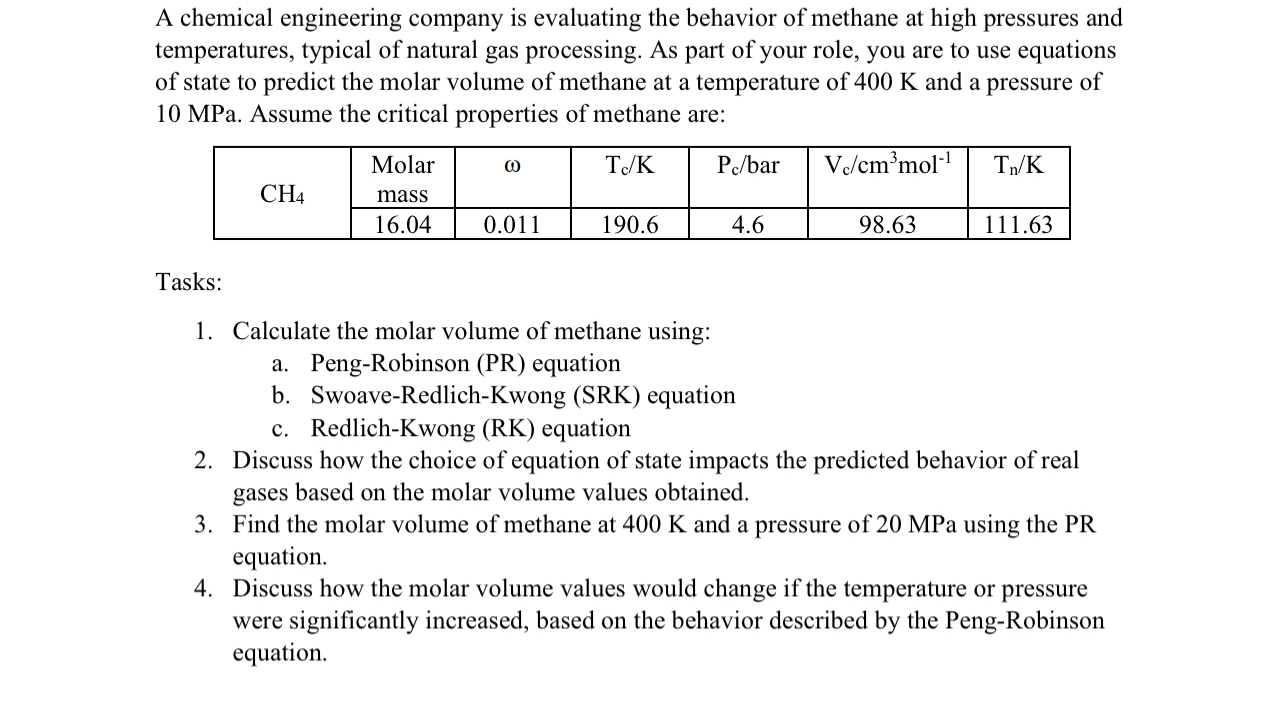
A chemical engineering company is evaluating the behavior of methane at high pressures and temperatures, typical of natural gas processing. As part of your role,
A chemical engineering company is evaluating the behavior of methane at high pressures and temperatures, typical of natural gas processing. As part of your role, you are to use equations of state to predict the molar volume of methane at a temperature of and a pressure of MPa. Assume the critical properties of methane are:
tabletableMolarmassbar
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


