Answered step by step
Verified Expert Solution
Question
1 Approved Answer
An air-SO2 gas mixture containing 3% SO2 by volume is contacted with water (B) containing 0.026% SO2 (A) by moles at 760 mmHg and 20C.
An air-SO2 gas mixture containing 3% SO2 by volume is contacted with water (B) containing 0.026% SO2 (A) by moles at 760 mmHg and 20C. Film mass transfer coefficients were calculated as kx =6.4*10-3 kmol/m2.s and ky =3.6*10-4 kmol/m2.s. The equilibrium data were obtained as follows.
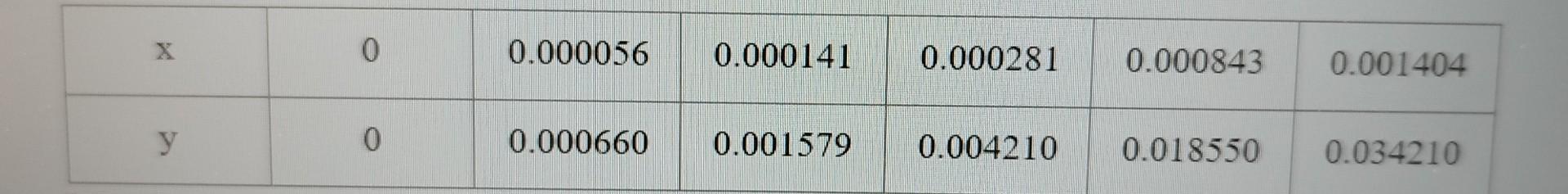
Accordingly; a) Interface concentrations, (b) the total molar flow of SO2; c) Calculate the resistance in the gas phase to mass transfer.
\begin{tabular}{|c|l|l|l|l|l|l|} \hline x & 0 & 0.000056 & 0.000141 & 0.000281 & 0.000843 & 0.001404 \\ \hline y & 0 & 0.000660 & 0.001579 & 0.004210 & 0.018550 & 0.034210 \\ \hline \end{tabular} \begin{tabular}{|c|l|l|l|l|l|l|} \hline x & 0 & 0.000056 & 0.000141 & 0.000281 & 0.000843 & 0.001404 \\ \hline y & 0 & 0.000660 & 0.001579 & 0.004210 & 0.018550 & 0.034210 \\ \hline \end{tabular}Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


