Answered step by step
Verified Expert Solution
Question
1 Approved Answer
ASAP. NO NEED TO SHOW FULL COMPUTATION, IAM JUST CHACKING MY WORK Calculate the G for the cell: Cu(s)Cu2+(0.010M)Ag+(0.050M)Ag(s). CuCu2+=0.337VAgAg+=0.799V 92.6kJ85.7kJ87.4kJ89.2kJ
ASAP. NO NEED TO SHOW FULL COMPUTATION, IAM JUST CHACKING MY WORK
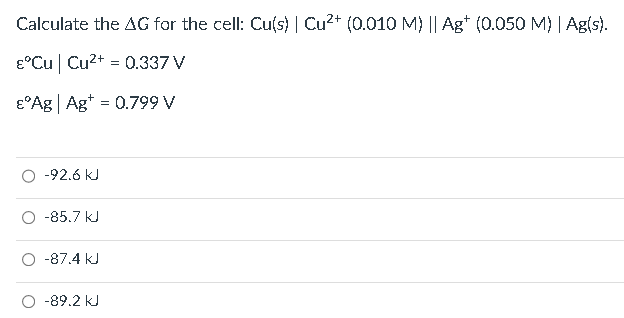
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


