Answered step by step
Verified Expert Solution
Question
1 Approved Answer
can I please get a step by step guide of solving these questions? Show your work - answers without supporting work will get zero marks.



can I please get a step by step guide of solving these questions?
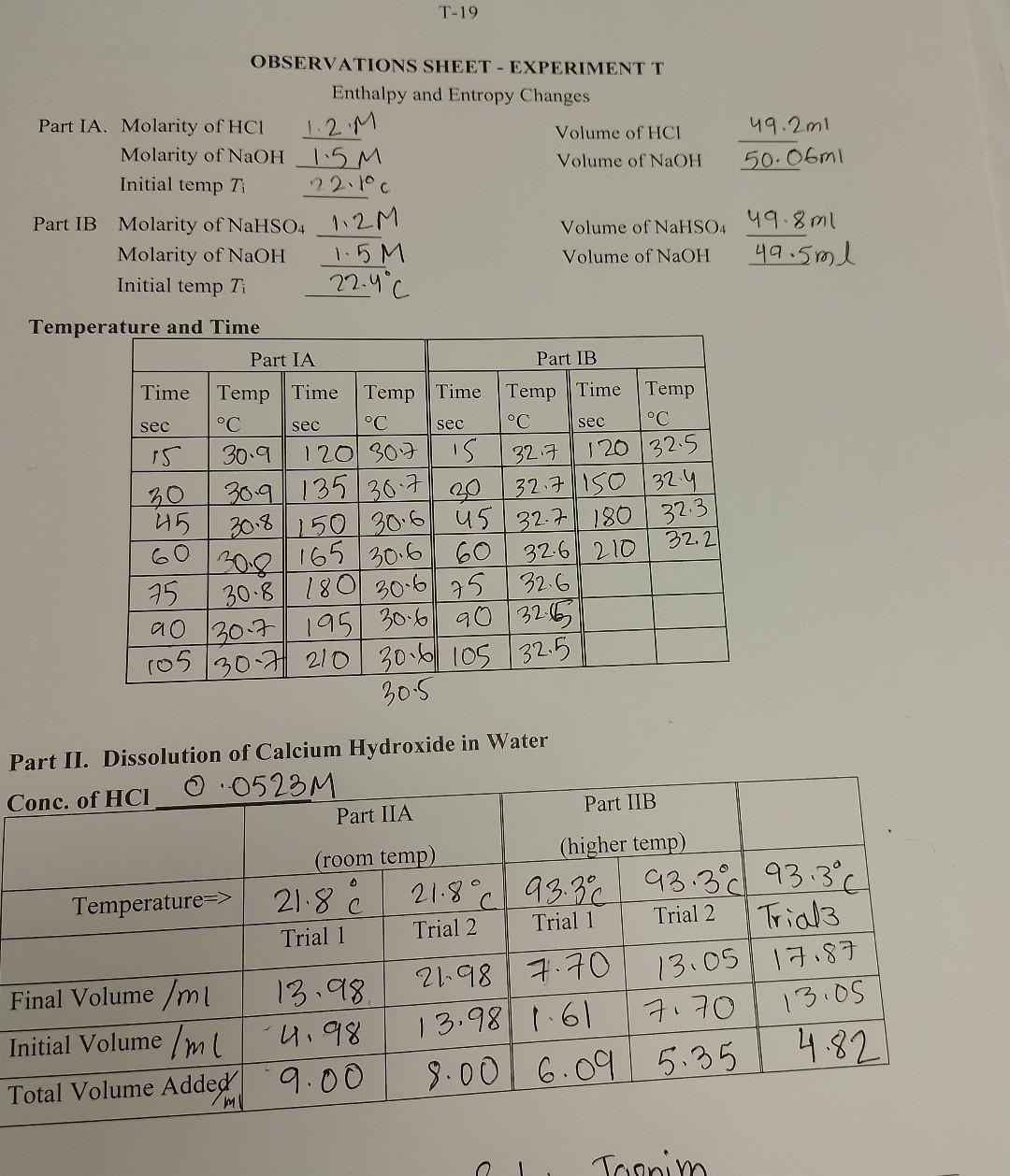



can I please get a step by step guide of solving these questions?

Show your work - answers without supporting work will get zero marks. Give units and use the correct number of significant figures in all calculations. A. Enthalnv of Neutralization of HCl : the graph for this part must be attached ( 2 marks). I. Enthalpy of Dissociation of an Acid Show below your calculation of Hneutralization in kJ/( mole of U1). B. Enthalpy of Neutralization of NaHSO4 Show below your calculation of HneutralizationinkJ/ (mole of NaHSU4). ( marks) C. Dissociation of NaHSO4 Calculate Hdissociation per mole of NaHSO4 in water. (1 mark) Part II Dissolution of Calcium Hydroxide in Water A. Show ONE calculation of Ksp. Report your average values of Ksp at each of the two temperatures. ( 1 mark) \begin{tabular}{ll} T(K)= & Ksp= \\ T(K)= & Ksp= \\ \hline \end{tabular} B. (a) Using the graph paper at the back of this manual, draw and attach a graph of lnKsp against 1/T (1 mark). (b) Calculate H in kJ/mol. (1.5 marks) (c) Calculate S in J/mol-degree. (1.5 marks) OBSERVATIONS SHEET - EXPERIMENT T Tempera Dart II. Dissolution of Calcium Hydroxide in Water
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


