Answered step by step
Verified Expert Solution
Question
1 Approved Answer
If an expert can please answer Part A and B questions by using calculations and showing your work. Also you must pay attention to the
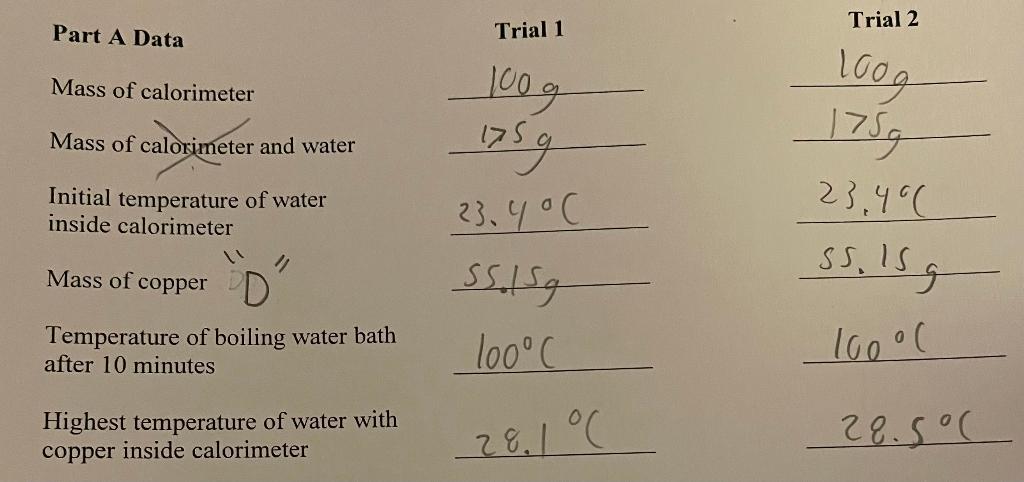

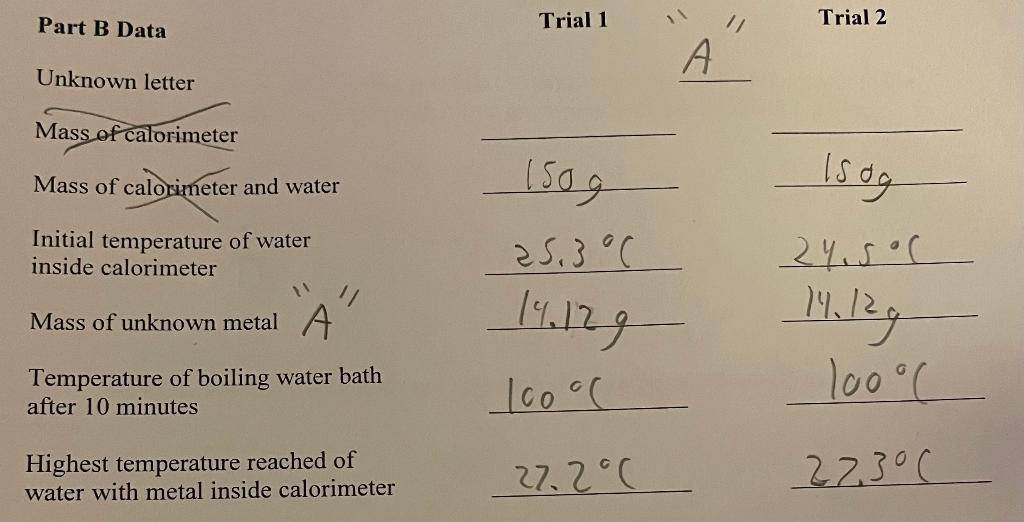

 If an expert can please answer Part A and B questions by using calculations and showing your work. Also you must pay attention to the significant figures. Also for question 5 from part B you must search up the table of select metals and their heat capacities. If an expert can please complete this as soon as possible I would really appreciate it please and thank you. This is for Chem 65.
If an expert can please answer Part A and B questions by using calculations and showing your work. Also you must pay attention to the significant figures. Also for question 5 from part B you must search up the table of select metals and their heat capacities. If an expert can please complete this as soon as possible I would really appreciate it please and thank you. This is for Chem 65.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


