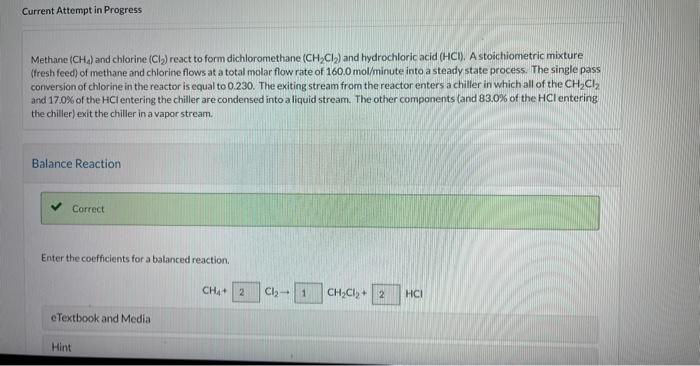
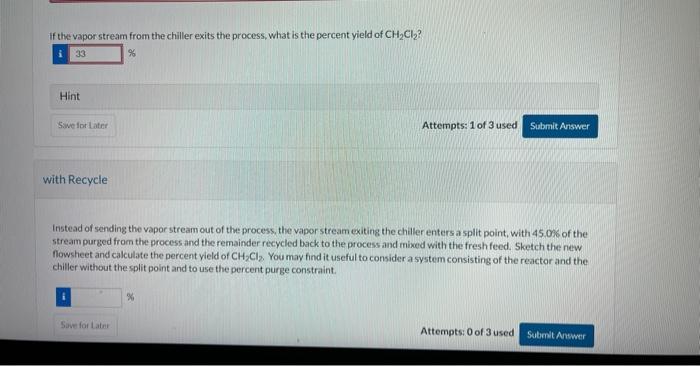
Methane (CH4) and chlorine (Cl2) react to form dichloromethane (CH2Cl2) and hydrochloric acid (HCl). A stoichiometric mixture (tresh feed) of methane and chlorine flows at a total molar flow rate of 160.0mol/ minute into a steady state process. The single pass conversion of chlorine in the reactor is equal to 0.230 . The exiting stream from the reactor enters a chiller in which all of the CH2Cl2 and 17.0% of the HCl entering the chiller are condensed into aliquid stream. The other components land 83.0% of the HCl entering the chiller) exit the chiller in a vapor stream. Balance Reaction Enter the coefficients for a balanced reaction. If the vapor stream from the chiller exits the process, what is the percent yield of CH2Cl2 ? Hint Attempts: 1 of 3 used with Recycle Instead of sending the vapor stream out of the process, the vapor stream exiting the chaler enters a split point, with 45.03 of the stream purged from the process and the remainder recycled back to the process and mixed with the fresh feed. Sketch the new. fowsheet and calculate the percent yield of CH2Cl2. You may find it useful to consider a system consisting of the reactor and the chiller without the split point and to use the percent purge constraint. Methane (CH4) and chlorine (Cl2) react to form dichloromethane (CH2Cl2) and hydrochloric acid (HCl). A stoichiometric mixture (tresh feed) of methane and chlorine flows at a total molar flow rate of 160.0mol/ minute into a steady state process. The single pass conversion of chlorine in the reactor is equal to 0.230 . The exiting stream from the reactor enters a chiller in which all of the CH2Cl2 and 17.0% of the HCl entering the chiller are condensed into aliquid stream. The other components land 83.0% of the HCl entering the chiller) exit the chiller in a vapor stream. Balance Reaction Enter the coefficients for a balanced reaction. If the vapor stream from the chiller exits the process, what is the percent yield of CH2Cl2 ? Hint Attempts: 1 of 3 used with Recycle Instead of sending the vapor stream out of the process, the vapor stream exiting the chaler enters a split point, with 45.03 of the stream purged from the process and the remainder recycled back to the process and mixed with the fresh feed. Sketch the new. fowsheet and calculate the percent yield of CH2Cl2. You may find it useful to consider a system consisting of the reactor and the chiller without the split point and to use the percent purge constraint