Answered step by step
Verified Expert Solution
Question
1 Approved Answer
PHYSICAL CHEMISTRY. NEED HELP!! Question 5 (19 Marks): Table 3 shows the equilibrium constants for the reaction 2A (9) + B (g) = 2C (g)
PHYSICAL CHEMISTRY. NEED HELP!! 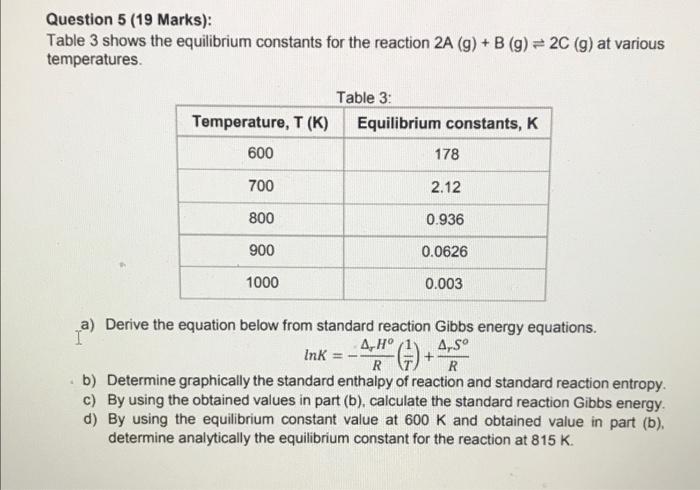
Question 5 (19 Marks): Table 3 shows the equilibrium constants for the reaction 2A (9) + B (g) = 2C (g) at various temperatures Table 3: Temperature, T (K) Equilibrium constants, K 600 178 700 2.12 800 0.936 900 0.0626 1000 0.003 a) Derive the equation below from standard reaction Gibbs energy equations. . (1) Ink * (75) + A. S R R b) Determine graphically the standard enthalpy of reaction and standard reaction entropy. C) By using the obtained values in part (b), calculate the standard reaction Gibbs energy d) By using the equilibrium constant value at 600 K and obtained value in part (b), determine analytically the equilibrium constant for the reaction at 815 K 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


