Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please explain each step thank you Reactive metals such as magnesium react readily with acids in aqueous solution. 2.17g of Mg is added to 81.4mL
please explain each step thank you 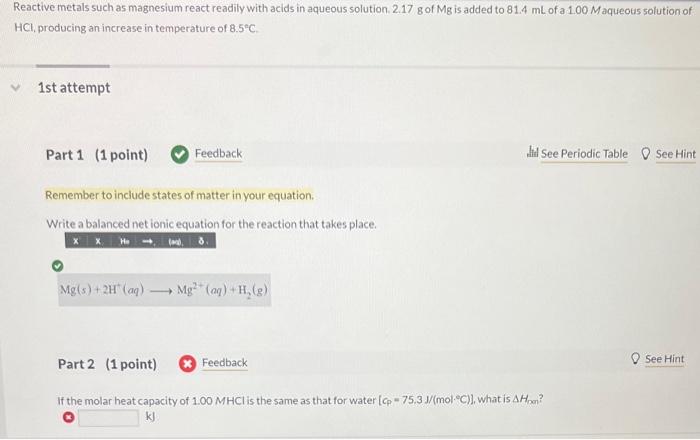
Reactive metals such as magnesium react readily with acids in aqueous solution. 2.17g of Mg is added to 81.4mL of a 1.00Maqueous solution of HCl, producing an increase in temperature of 8.5C. 1st attempt Part 1 (1 point) Feedback Wit See Periodic Table 0 See Hint Remember to include states of matter in your equation. Write a balanced net ionic equation for the reaction that takes place. Mg(s)+2H+(aq)Mg2+(aq)+H2(g) Part 2 (1 point) Feedback If the molar heat capacity of 1.00MHCl is the same as that for water [cp=75.3J/(molC)], what is Hpen ? kJ 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


