Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please help, i'm so lost... Nitric oxide reacts with chlorine gas according to the following reaction: 2NO(g)+Cl2(g)2NOCl(g)2NO(g)+Cl2(g)2NOCl(g) Kp=0.27p=0.27 at 700 K A reaction mixture initially
please help, i'm so lost...
Nitric oxide reacts with chlorine gas according to the following reaction: 2NO(g)+Cl2(g)2NOCl(g)2NO(g)+Cl2(g)2NOCl(g) Kp=0.27p=0.27 at 700 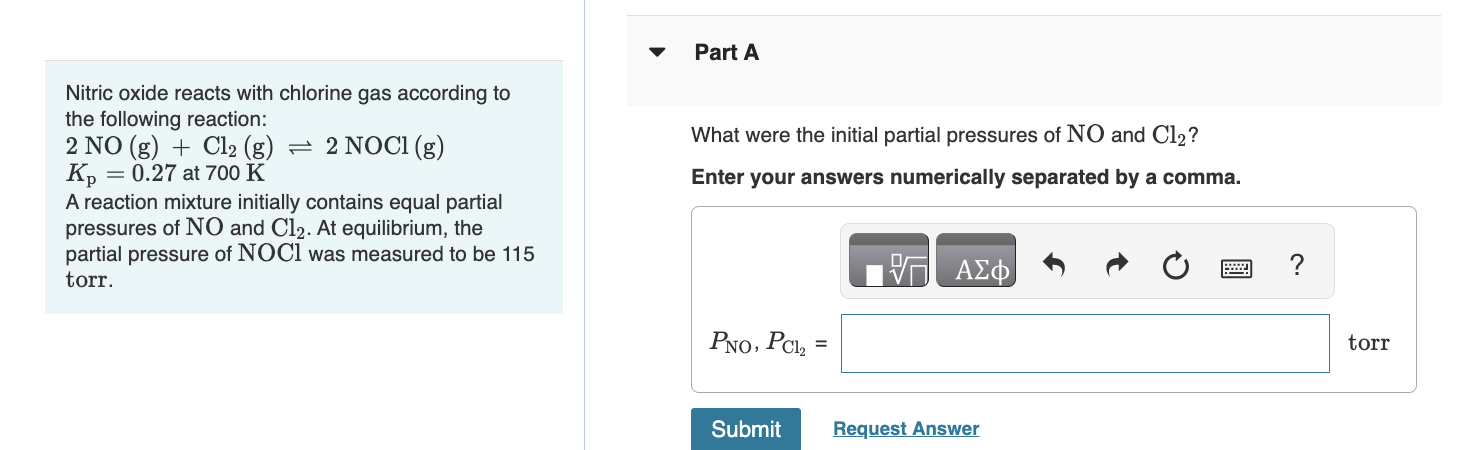 K A reaction mixture initially contains equal partial pressures of NONO and Cl2Cl2. At equilibrium, the partial pressure of NOClNOCl was measured to be 115 torrtorr.
K A reaction mixture initially contains equal partial pressures of NONO and Cl2Cl2. At equilibrium, the partial pressure of NOClNOCl was measured to be 115 torrtorr.
What were the initial partial pressures of NONO and Cl2Cl2?Enter your answers numerically separated by a comma.
PNO, PCl2 = __ torr
Nitric oxide reacts with chlorine gas according to the following reaction: 2NO(g)+Cl2(g)2NOCl(g)Kp=0.27at700K What were the initial partial pressures of NO and Cl2 ? Enter your answers numerically separated by a comma. A reaction mixture initially contains equal partial pressures of NO and Cl2. At equilibrium, the partial pressure of NOCl was measured to be 115 torrStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


