Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Please show all work and explain it. Thank you A cylindrical tank of known dimensions containing m=4kg of carbon monoxide gas (molar mass is M=28g/mc
Please show all work and explain it.
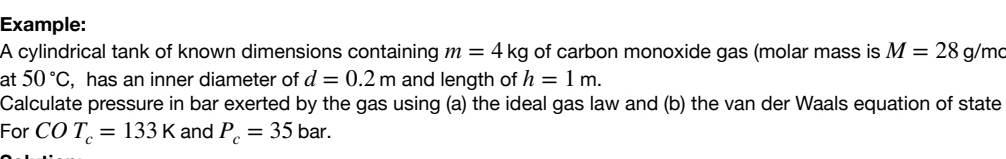
Thank you
A cylindrical tank of known dimensions containing m=4kg of carbon monoxide gas (molar mass is M=28g/mc at 50C, has an inner diameter of d=0.2m and length of h=1m. Calculate pressure in bar exerted by the gas using (a) the ideal gas law and (b) the van der Waals equation of state oor COTc=133K and Pc=35barStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


