Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Made of high-strength steel, a tie rod is a slender cylindrical structure used to push (compression) and pull (tension) the front tires when the
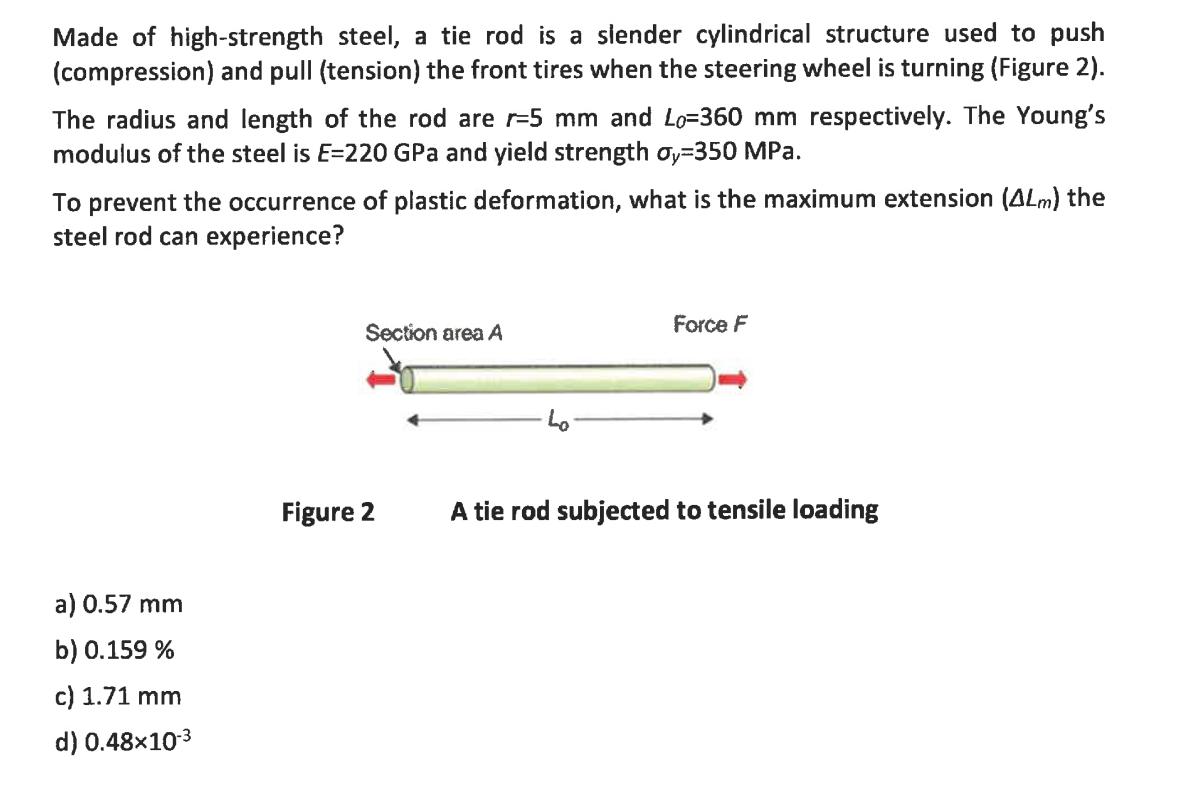

Made of high-strength steel, a tie rod is a slender cylindrical structure used to push (compression) and pull (tension) the front tires when the steering wheel is turning (Figure 2). The radius and length of the rod are r-5 mm and Lo=360 mm respectively. The Young's modulus of the steel is E=220 GPa and yield strength oy-350 MPa. To prevent the occurrence of plastic deformation, what is the maximum extension (4Lm) the steel rod can experience? a) 0.57 mm b) 0.159 % c) 1.71 mm d) 0.48x10-3 Section area A Figure 2 Force F A tie rod subjected to tensile loading Please select the FALSE statement a) In a crystalline metallic structure, the atoms are arranged in a three-dimensional array called a lattice. b) A BCC unit cell consists of eight atoms at the corners of a cube and one atom at the body centre of the cube. c) Hydrogen bond is a type of bond, which occurs due to the ease with which hydrogen atoms are willing to give up an electron to atoms of oxygen, fluorine, or nitrogen (x) d) FCC crystal structures are more close-packed than BCC crystal structures.
Step by Step Solution
★★★★★
3.46 Rating (153 Votes )
There are 3 Steps involved in it
Step: 1
To find the maximum extension L without plastic deformation we can use Hookes Law which states that ...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


