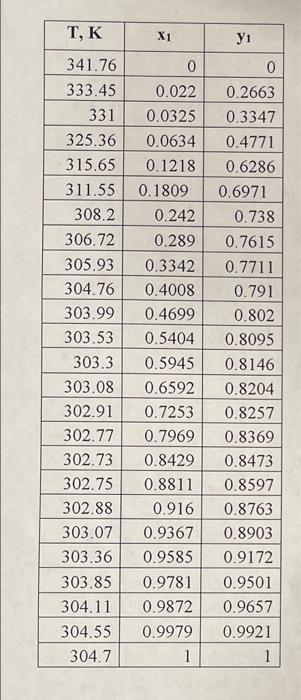
Problem 2. A liquid containing 40 mole % methyl methanoate and 60 mole % hexane is continuously fed to and distilled in a flash drum at 101.32kPa. The feed rate is 100 kg moles/hr. Equilibrium data is given in Table 1. Print the operating lines from parts (a) - (c) on the same diagram and label each line. Printing the diagram and labeling each operating line by hand is allowed. a. If 61% of the feed remains as liquid, what are the vapor and liquid mole fractions for benzene, and the total vapor (V) and liquid (L) flow rates? Show the operating line on the xy diagram and plot the diagram in mole fractions of benzene. Show the equation for the operating line with numerical values for the slope and the intercept. b. If the desired liquid product is 10 mole % methyl methanoate, what vapor fraction (V/F) should be used? Determine the total product flow rates (V and L) and mole fraction of methyl methanoate in the vapor phase. Show the operating line on the xy diagram. c. If the drum temperature is 306.72K, find the mole fractions of methyl methanoate in the liquid and vapor products (using Txy data) and calculate V/F. Show the operating line on the xy diagram. \begin{tabular}{|r|r|r|} \hline \multicolumn{1}{|c|}{ T, K } & \multicolumn{1}{c|}{x1} & \multicolumn{1}{c|}{y1} \\ \hline 341.76 & 0 & 0 \\ \hline 333.45 & 0.022 & 0.2663 \\ \hline 331 & 0.0325 & 0.3347 \\ \hline 325.36 & 0.0634 & 0.4771 \\ \hline 315.65 & 0.1218 & 0.6286 \\ \hline 311.55 & 0.1809 & 0.6971 \\ \hline 308.2 & 0.242 & 0.738 \\ \hline 306.72 & 0.289 & 0.7615 \\ \hline 305.93 & 0.3342 & 0.7711 \\ \hline 304.76 & 0.4008 & 0.791 \\ \hline 303.99 & 0.4699 & 0.802 \\ \hline 303.53 & 0.5404 & 0.8095 \\ \hline 303.3 & 0.5945 & 0.8146 \\ \hline 303.08 & 0.6592 & 0.8204 \\ \hline 302.91 & 0.7253 & 0.8257 \\ \hline 302.77 & 0.7969 & 0.8369 \\ \hline 302.73 & 0.8429 & 0.8473 \\ \hline 302.75 & 0.8811 & 0.8597 \\ \hline 302.88 & 0.916 & 0.8763 \\ \hline 303.07 & 0.9367 & 0.8903 \\ \hline 303.36 & 0.9585 & 0.9172 \\ \hline 303.85 & 0.9781 & 0.9501 \\ \hline 304.11 & 0.9872 & 0.9657 \\ \hline 304.55 & 0.9979 & 0.9921 \\ \hline 304.7 & 1 & 1 \\ \hline \end{tabular}